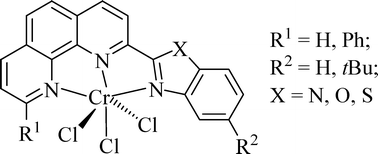
The 2-benzazole-1,10-phenanthrolines and their chromium(III) complexes were synthesized and characterized by elemental and spectroscopic analyses as well as by single-crystal X-ray crystallography. X-Ray crystallographic analyses of C6 and C8 reveal an octahedral geometry around both chromium centers. The electronic spectra of the chromium complexes exhibit d–d transitions typically of a pseudo-octahedral coordinated d3 ion, falling into the region ν14A2g→4T2g 600–700 nm,ν24A2g→4T1g(F) 430–470 nm, and the 10Dq values between 15674 and 16529 cm−1. The variation of the heteroatoms (X = N, O, S) in benzazole group shows regular influence on the 10Dq values as 10Dq (O) > 10Dq (N) > 10Dq (S), which reflects the σ-donor/π-acceptor properties of these benzazole ligands. Upon activation with modified methylaluminoxane (MMAO), these complexes exhibited high activities for ethylene oligomerization (up to 7.36 × 106 g mol−1 (Cr) h−1) and ethylene polymerization (up to 1.28 × 106 g mol−1 (Cr) h−1). The effects of non-coordinated N, O or S heteroatoms in the ligands have been examined, and interestingly catalytic activities also strictly adhered to the trend O > N > S under various ethylene pressures. Reaction conditions significantly influenced the catalytic properties of the complexes.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...