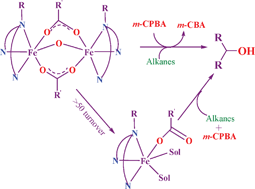
A few diiron(III) complexes [Fe2(O)(OAc)2(L1)2](ClO4)21, [Fe2(O)(OBz)2(L1)2](ClO4)22, [Fe2(O)(OAc)2(L2)2](ClO4)23 and [Fe2(O)(OBz)2(L2)2](ClO4)24, where L1 = N,N-bis(pyrid-2-ylmethyl)-iso-butylamine, L2 = N,N-bis(pyrid-2-ylmethyl)benzylamine, AcO = acetate and BzO = benzoate, have been isolated and characterized by means of elemental analysis and spectral and electrochemical methods. The molecular structures of the complexes 2 and 4 have been determined by single-crystal X-ray diffraction analysis and they possess a distorted bioctahedral geometry in which each iron atom is coordinated to the oxygen atom of the μ-oxo bridge, two oxygen atoms of the μ-benzoato bridges and three nitrogen atoms of L1 and L2 ligands capping the two ends of the diiron(III) cluster. The ESI-MS spectral data of the complexes reveal that the complexes remain intact in dichloromethane (DCM) solution. Upon adding one equivalent of Et3N to a mixture of one equivalent of the diiron(III) complexes and excess of m-chloroperbenzoic acid (m-CPBA) in DCM, an intense absorption band (λmax, 670–700 nm) appears, which corresponds to the species [Fe2(O)(OAc)(m-CPBA)(L)2]2+ (ESI-MS, m/z 466) suggested as the intermediate involved in the oxygenation reactions. All the present complexes show efficient alkane hydroxylation with 300–400 turn over numbers and good selectivities for cyclohexane (A/K, 10–14) and adamantane (3°/2°, 9–11). Interestingly, the formation of monoiron(III) species has been discerned in the alkane hydroxylation reactions beyond ∼50 turnovers. The mononuclear 1 : 1 iron(III) complexes of L1 and L2 ligands generated in situ are also found to catalyze the oxygenation reactions with high selectivity and efficiency for cyclohexane (A/K, 10–14). Upon their reaction with m-CPBA in DCM, a characteristic absorption band (λmax, 600 nm, εmax, 355 M−1 cm−1) appears and decays at room temperature. This spectral feature is consistent with the mononuclear high-valent iron-oxo species suggested as an intermediate in the oxygenation reactions.

 Please wait while we load your content...
Please wait while we load your content...