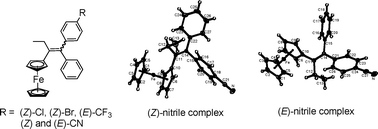
We have been exploring the cytotoxic effects of conjugated phenylferrocene systems on breast cancer cells. Complexes with p-OH, p-NH2, and p-NHC(O)CH3 substitution show particularly high activity, with IC50 values in the low or sub micromolar range for both the hormone-dependent MCF-7 and hormone-independent MDA-MB-231 breast cancer cell lines. We now present the synthesis, X-ray crystal structures and biochemical studies of analogous halogen or pseudo-halogenpara-substituted compounds with R = Cl, (Z)-7a; Br, (Z)-7b; CF3, (E)-7c; and CN, (E)-7d and (Z)-7d. Lacking hydrogen bonding groups, the compounds have low, but non-zero, relative binding affinity values for the oestrogen receptor alpha (RBA ≤ 0.55%) as well as mildly exothermic ligand binding in in silicoER docking experiments. All compounds show estrogenic (proliferative) activity on the MCF-7 cell line. On MDA-MB-231 cells, the cyano complex (Z)-7d shows a reasonable cytotoxic effect (IC50 = 11 μM), its isomer (E)-7d is only slightly cytotoxic (IC50 = 60 μM), while the Cl, Br, and CF3 derivatives have no effect. Cytotoxic properties, while they correlate somewhat with the resonance donating abilities of the substituent, are more strongly dependent on the presence of a proton in the functional group, supporting our prior proposition that electrophilic quinoid forms of the compounds could be active species in the cell. A correlation of the redox potential of the ferrocenyl moiety with the Hammett-Taft constants of the substituents was observed.

 Please wait while we load your content...
Please wait while we load your content...