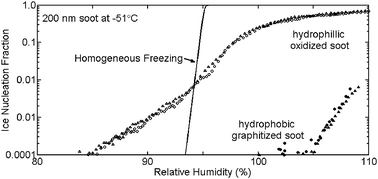
Cloud condensation nuclei (CCN) activity and ice nucleation behavior (for temperatures ≤−40 °C) of soot aerosols relevant for atmospheric studies were investigated. Soots were chosen to represent a range of physico-chemical properties, from hydrophobic through a range of hydrophilicity, to hygroscopic. These characteristics were achieved through generation by three different combustion sources; three soots from natural gas pyrolysis (original: TS; graphitized: GTS; and oxidized: TOS), soot from a diffusion flame in an oil lamp burning aviation kerosene (TC1), and soot from a turbulent diffusion flame in an aircraft engine combustor (AEC). All of the samples exhibited some heterogeneity in our experiments, which showed evidence of two or more particle sub-types even within a narrow size cut. The heterogeneity could have resulted from both chemical and sizing differences, the latter attributable in part to particle non-sphericity. Neither GTS nor TS, hydrophobic particles distinguished only by the lower porosity and polarity of the GTS surface, showed CCN activity at or below water supersaturations required for wettable, insoluble particles (the Kelvin limit). TC1 soot particles, despite classification as hydrophilic, did not show CCN activity at or below the Kelvin limit. We attribute this result to the microporosity of this soot. In contrast, oxidized, non-porous, and hydrophilic TOS particles exhibited CCN activation at very near the Kelvin limit, with a small percentage of these particles CCN-active even at lower supersaturations. Due to containing a range of surface coverage of organic and inorganic hydrophilic and hygroscopic compounds, up to ∼35% of hygroscopic AEC particles were active as CCN, with a small percentage of these particles CCN-active at lower supersaturations. In ice nucleation experiments below −40 °C, AEC particles nucleated ice near the expected condition for homogeneous freezing of water from aqueous solutions. In contrast, GTS, TS, and TC1 required relative humidity well in excess of water saturation at −40 °C for ice formation. GTS particles required water supersaturation conditions for ice activation even at −51 °C. At −51 to −57 °C, ice formation in particles with electrical mobility diameter of 200 nm occurred in up to 1 in 1000 TS and TC1 particles, and 1 in 100 TOS particles, at relative humidities below those required for homogeneous freezing in aqueous solutions. Our results suggest that heterogeneous ice nucleation is favored in cirrus conditions on oxidized hydrophilic soot of intermediate polarity. Simple considerations suggest that the impact of hydrophilic soot particles on cirrus cloud formation would be most likely in regions of elevated atmospheric soot number concentrations. The ice formation properties of AEC soot are reasonably consistent with present understanding of the conditions required for aircraft contrail formation and the proportion of soot expected to nucleate under such conditions.

 Please wait while we load your content...
Please wait while we load your content...