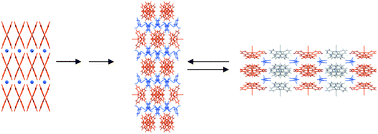
Crystal disassembly and reassembly processes are exploited to develop more efficient routes to known compounds [CuI(phen)2)]I·H2O (2) and [CuI(phen)2)] [(PhPO(OH)2)·(PhP(O)2OH)] (3), and routes to novel compounds such as [CuI(phen)2)][(PhS(O)3)]·2H2O (4), a novel sulfonate analogue of (3). Previous attempts to produce (2) have involved 18 hour reflux of ethanolic solutions of [Cu(phen)I]. Here the same material was produced by oxidation of [Cu(I)(phen)2]I (1) with iodine for 20 min at 70 °C. Compound (3), which is generally prepared by oxidation of (1) with phenylphosphonic acid, was successfully obtained, and in significantly greater yield, by reaction of (2) with excess phenylphosphonic acidi.e. displacement of the iodide counter-ion by a phenylphosphonate dimer anion (Route A, Scheme 1). The novel sulfonate analogue (4) was formed by displacement of the phenylphosphonate dimer anion in (3) by a phenylsulfonate anion (Route B). This reaction also proved reversible. Thus, both routes demonstrate the mobility of the counter-anions (2), (3) and (4). However, attempts to produce sulfonic and arsonic acid analogues by Route A, and arsonic acid analogues by Route B, have proved unsuccessful to-date. Compounds (3) and (4) exhibit similar crystal class (both monoclinic), but the former is c-centred whilst the latter is primitive. The general assembly pattern of ‘ribbons’ of [Cu(II)(phen)2I]+ cations interleaved with sheets of acid anions seen in (3), is maintained to some extent in the sulfonate analogue (4). However, the discrete sulfonate anions in (4) (not dimers as in (3)) rely principally on H-bonding interactions with water molecules in order to form sheets.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...