Highly stereoselective intramolecular α-arylation of self-stabilized non-racemic enolates: synthesis of α-quaternary α-amino acid derivatives†
Abstract
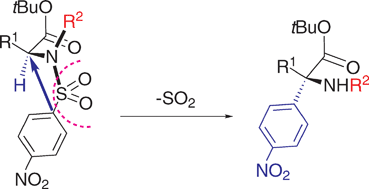
The ‘one-pot’ stereoselective conversion of N-(4-nitrobenzene)sulfonyl-α-amino acid tert-butyl esters into the corresponding N-alkyl-α-(4-nitrophenyl)-α-amino esters has been realized through

 Please wait while we load your content...
Please wait while we load your content...