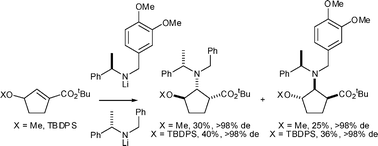
tert-Butyl (RS)-3-methoxy- and (RS)-3-tert-butyldiphenylsilyloxy-cyclopent-1-ene-carboxylates display excellent levels of enantiorecognition in mutual kinetic resolutions with both lithium (RS)-N-benzyl-N-(α-methylbenzyl)amide and lithium (RS)-N-3,4-dimethoxybenzyl-N-(α-methylbenzyl)amide. A 50 : 50 pseudoenantiomeric mixture of lithium (S)-N-benzyl-N-(α-methylbenzyl)amide and lithium (R)-N-3,4-dimethoxybenzyl-N-(α-methylbenzyl)amide allows for the efficient parallel kinetic resolution of the tert-butyl (RS)-3-oxy-substituted cyclopent-1-ene-carboxylates, affording differentially protected 3-oxy-substituted cispentacin derivatives in high yield and >98% de. Subsequent N-deprotection and hydrolysis provides access to 3-oxy-substituted cispentacin derivatives in good yield, and in >98% de and >98% ee, while stereoselective epimerisation and subsequent deprotection affords the corresponding transpentacin analogues in good yield, and in >98% de and >98% ee.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...