The coordination chemistry of a series of potentially tetra-, tri- and bi-dentate aminophenol pro-ligands ([L1]H–[L5]H) with magnesium and zinc derivatives has been studied. Reactions of the pro-ligands [L1]H, [L2]H and [L3]H with one equiv. of ZnEt2 or Zn(N(SiMe3)2)2 in toluene at room temperature afford cleanly, viaethane or amine elimination, the ethyl- and amido-zinc complexes 1–4. Complexes [L4]ZnEt (5) and [L4]Zn(N(SiMe3)2)2 (6) derived from the tridentate pro-ligand [L4]H were prepared in 84% and 76% yields, following similar alkane and amine elimination protocols, respectively. The 1 : 1 reactions of bidentate pro-ligand [L5]H with ZnEt2 or Zn(N(SiMe3)2)2 in toluene using different reaction protocols systematically yielded mixtures of the bis(ligand) complex [L5]2Zn (7) and the corresponding ethyl-{[L5]ZnEt}n (8; n = 1 or 2) or amido- {[L5]Zn(N(SiMe3)2)}n complexes (9; n = 1 or 2). The synthesis of magnesium-enolate complexes 10 and 11 was carried out in a one-pot, two-step procedure by first reacting pro-ligands [L1]H and [L4]H with one equiv. of Mg(n,sBu)2 to generate the corresponding {[Ln]Mg(n,sBu)}n species, which were further reacted with one equiv. of 2,4,6-Me3C6H2COMe. All complexes have been characterized by multinuclear NMR, elemental analysis, and by single-crystal X-ray diffraction studies for five-coordinate magnesium complex 10, and four-coordinate Zn complexes 3–7. Preliminary studies indicate that magnesium species 10 and 11 as well as zinc complexes 1 and 5 are not active initiators for the polymerization of methyl methacrylate, even in combination with one equiv. of Li[OC(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2)(2,4,6-Me3C6H2)], while zinc complexes 2, 4 and 6 are effective initiators for the ring-opening polymerization of ε-caprolactone and rac-lactide at 20 °C.
CH2)(2,4,6-Me3C6H2)], while zinc complexes 2, 4 and 6 are effective initiators for the ring-opening polymerization of ε-caprolactone and rac-lactide at 20 °C.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2)(2,4,6-Me3C6H2)], while zinc complexes 2, 4 and 6 are effective initiators for the ring-opening polymerization of
CH2)(2,4,6-Me3C6H2)], while zinc complexes 2, 4 and 6 are effective initiators for the ring-opening polymerization of 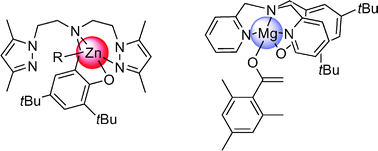

 Please wait while we load your content...
Please wait while we load your content...