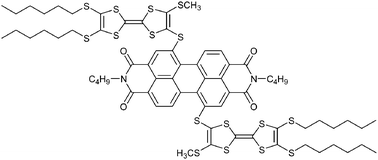
A donor–acceptor–donor dyad 4 involving 2-sulfur-3-methylthio-6,7-bis(hexylthio)tetrathiafulvalene (TTF) as a donor attached directly to N,N′-dibutylperylene-3,4,9,10-tetracarboxylic diimide (PDI) as an acceptor was synthesized by condensation of N,N′-dibutyl-1,6-dibromo-3,4,9,10-perylenetetracarboxylic diimide and 2-(2-cyanoethylthio-3-methylthio-6,7-bis(hexylthio)tetrathiafulvalene. The cyclic voltammetric (CV) data implied significant intramolecular interaction and the absorption spectrum indicated that there was an intramolecular charge transfer (ICT) interaction between TTF and PDI moieties in dyad 4. Comparing with PDI 13, the fluorescence emission intensity of dyad 4 was quenched almost quantitatively, which might result from the photo-induced electron transfer (PET) interaction between the PDI and TTF moieties in dyad 4. The fluorescence intensity of dyad 4 could be reversibly modulated by sequential oxidation and reduction of the TTF unit using chemical methods. Thus dyad 4 can be regarded as a new reversible fluorescence-redox dependent molecular switch.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...