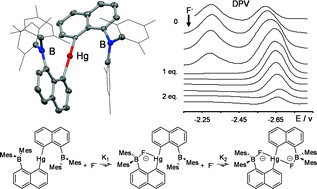
The reaction of [Li(THF)4][1,8-μ-(Mes2B)C10H6] with HgCl2 affords [1,1′-(Hg)-[8-(Mes2B)C10H6]2] (3) or [1-(ClHg)-8-(Mes2B)C10H6] (4), depending on the stoichiometry of the reagents. These two new compounds have been characterized by 1H, 13C, 11B and 199Hg NMR, elemental analysis and X-ray crystallography. The cyclic voltammogram of 3 in THF shows two distinct waves observed at E1/2 −2.31 V and −2.61 V, corresponding to the sequential reductions of the two boron centers. Fluoride titration experiments monitored by electrochemistry suggest that 3 binds tightly to one fluoride anion and more loosely to a second one. Theses conclusions have been confirmed by a UV-vis titration experiment which indicates that the first fluoride binding constant (K1) is greater than 108 M−1 while the second (K2) equals 5.2 (± 0.4) × 103 M−1. The fluoride binding properties of 3 have been compared to those of [1-(Me2B)-8-(Mes2B)C10H6] (1) and [1-((2,6-Me2-4-Me2NC6H2)Hg)-8-(Mes2B)C10H6] (2). Both experimental and computational results indicate that its affinity for fluoride anions is comparable to that of 2 but significantly lower than that of the diborane 1. In particular, the fluoride binding constants of 1, 2 and 3 in chloroform are respectively equal to 5.0 (± 0.2) × 105 M−1, 1.0 (± 0.2) × 103 M−1 and 1.7 (± 0.1) × 103 M−1. Determination of the crystal structures of the fluoride adducts [S(NMe2)3][1–μ2-F] and [S(NMe2)3][3–μ2-F] along with computational results indicate that the higher fluoride binding constant of 1 arises from a strong chelate effect involving two fluorophilic boron centers.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...