Syntheses, structures and properties of seven isomorphous 1D Ln3+complexes Ln(BTA)(HCOO)(H2O)3 (H2BTA = bis(tetrazoly)amine, Ln = Pr, Gd, Eu, Tb, Dy, Er, Yb) and two 3D Ln3+ complexes Ln(HCOO)3 (Ln = Pr, Nd)†
Abstract
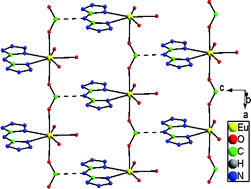
Seven isomorphous 1D chain Ln3+ complexes Ln(BTA)(HCOO)(H2O)3 (Ln = Pr (1), Gd (2), Eu (3), Tb (4) Dy (5), Er (6) and Yb (7)), and two formate coordinating and bridging 3D Ln3+ complexes Ln(HCOO)3 (Ln = Pr (8) and Nd (9)) have been synthesized and characterized by

 Please wait while we load your content...
Please wait while we load your content...