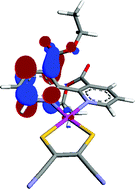
A series of [Pt(II)(diimine)(dithiolate)] complexes of general formula [Pt{X,X′-(CO2R)2-bpy}(mnt)] (where X = 3, 4 or 5; R = H or Et, bpy = 2,2′-bipyridyl and mnt = maleonitriledithiolate), have been spectroscopically, electrochemically and computationally characterised and compared with the precursors [Pt{X,X′-(CO2R)2-bpy}Cl2] and X,X′-(CO2R)2-bpy. The study includes cyclic voltammetry, in situEPR spectroelectrochemical studies of fluid solution and frozen solution samples, UV/Vis/NIR spectroelectrochemistry, hyrid DFT and TD-DFT calculations. The effect of changing the position of the bpy substituents from 3,3′ to 4,4′ and 5,5′ is discussed with reference to electronic changes seen within the different members of the family of molecules. The performance of the mnt complexes in dye-sensitised solar cells has been previously described and the superior performance of [Pt{3,3′-(CO2R)2-bpy}(mnt)] is now explained in terms of decreased electronic delocalisation through twisting of the bipyridyl ligand as supported by the EPR and computational results.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...