Formation and dissolution of metals are two of the oldest technical chemical processes. On the atomic scale, these processes are based on the formation and cleavage of metal–metal bonds. During the past 15 years we have studied intensively the intermediates during the formation process of metals, i.e. the formation of compounds containing many metal–metal bonds between naked metal atoms in the center and ligand-bearing metal atoms at the surface. We have called the clusters metalloid or, more generally, elementoid clusters. Via a retrosynthetic route, the many different Al and Ga metalloid clusters which have been structurally characterized allow us to understand also the dissolution process; i.e. the cleavage of metal–metal (M–M) bonds. However, this process can be detected much more directly by the reaction of single metal atom clusters in the gas phase under high vacuum conditions. A suitable tool to monitor the dissolution process of a metal cluster in the gas phase is FT-ICR (Fourier transform ion cyclotron resonance) mass spectrometry. Snapshots during these cleavage processes are possible because only every 1–10 s is there a contact between a cluster molecule and an oxidizing molecule (e.g. Cl2). This period is long, i.e. the formation of the primary product (a smaller metal atom cluster) is finished before the next collision happens. We have studied three different types of reaction:
(1) Step-by-step fragmentation of a structurally known metalloid cluster allows us to understand the bonding principle of these clusters because in every step only the weakest bond is broken.
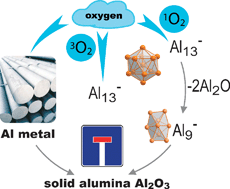
(2) There are three oxidation reactions of an Al13− cluster molecule with Cl2, HCl and O2 central to this review. These three reactions represent three different reaction types, (a) an exothermic reaction (Cl2), (b) an endothermic reaction (HCl), and (c) a kinetically limited reaction based on spin conservation rules (O2).
(3) Finally, we present the reaction of a metalloid cluster with Cl2 in order to show that in this cluster only the central naked metal atoms are oxidized, and a smaller metalloid cluster results containing the entire protecting shell as the primary cluster.
All the experimental results, supported by quantum chemical calculations, give a rough idea about the complex reaction cascades which occur during the dissolution and formation of metals. Furthermore, these results cast a critical light on many simplifying and generalizing rules in order to understand the bonding and structure of metal clusters. Finally, the experiments and some recent results provided by physical measurements on a crystalline Ga84 compound build a bridge to nanoscience; i.e. they may be a challenge for chemistry in the next decades, since it has been shown that only with a perfect orientation of nanoscale metal clusters, e.g. in a crystal, can novel, unexpected properties (e.g. superconducting nanoscale materials) be obtained.

 Please wait while we load your content...
Please wait while we load your content...