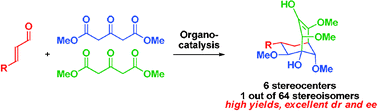
Controlling the formation of 1 out of 64 stereoisomers using organocatalysis†
Abstract
The formation of 1 out of 64 stereoisomers, i.e. controlling the creation of 6 stereocenters, of important optically active bicyclo[3.3.1]non-2-ene compounds, has been achieved using organocatalysis; the reaction proceeds with excellent stereocontrol and can be carried out with the generation of enantiopure products using

 Please wait while we load your content...
Please wait while we load your content...