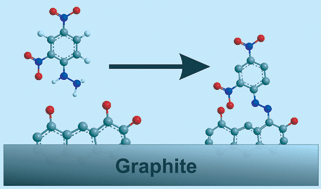
Quinone groups on the surface of edge-plane and basal-plane pyrolytic graphite electrodes, and, for the first time, “bamboo-like” multi-walled carbon nanotubes (b-MWCNTs), are labelled with a voltammetric “tag”, namely 2,4-dinitrophenylhydrazine (2,4-DNPH). The voltammetric response of these materials is studied in both aqueous and non-aqueous electrolyte solutions, and mechanisms are proposed for each media involving the voltammetrically controlled cleavage of the 2,4-DNPH adduct. Thus, these materials can be used for potentiometrically controlled chemical release, and as such may find application in, for example, drug delivery systems. In aqueous media, this cleavage results in the conversion of the surface quinonyl groups into surface arylnitroso species. This conversion is confirmed by studying the voltammetric response of the arylnitroso/arylhydroxylamine redox couple, formed after cleavage of the 2,4-DNPH adduct in the presence of L-glutathione, which has been shown in our previous work to react with arylnitroso species on the surface of graphitic carbon materials.

 Please wait while we load your content...
Please wait while we load your content...