Synthesis, computational and electrochemical characterization of a family of functionalized dimercaptothiophenes for potential use as high-energy cathode materials for lithium/lithium-ion batteries†‡
Abstract
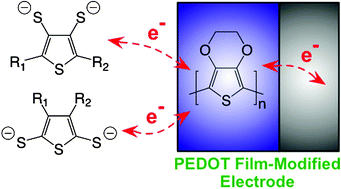
We present a family of a novel class of organosulfur compounds based on dimercaptothiophene and its derivatives, with a variety of functional groups (electron-donating or electron-withdrawing groups) and regiochemistries, designed as potential high-energy cathode materials with sufficient charge/discharge cyclability for lithium/lithium-ion rechargeable batteries. This study uses as a point of departure the electrochemical and computational understanding of the electrocatalytic effect of poly(3,4-ethylenedioxythiophene) (PEDOT) towards the redox reactions of 2,5-dimercapto-1,3,4-thiadiazole (DMcT). The effective redox potentials of these materials exhibited good correlation with the highest-occupied molecular orbital (HOMO) levels predicted via computational modeling. Furthermore, the redox reactions of all the compounds studied were electrocatalytically accelerated at PEDOT film-coated glassy carbon electrodes (GCEs), although some materials exhibited higher energy output than others. By using this approach we have identified several compounds that exhibit clear promise as potential cathode materials and have characterized the molecular interactions between the organosulfur compounds and PEDOT film surfaces involved in the electrocatalytic reactions.

 Please wait while we load your content...
Please wait while we load your content...