A facile pathway to fabricate microcapsules by in situ polyelectrolyte coacervation on poly(styrene sulfonate)-doped CaCO3 particles
Abstract
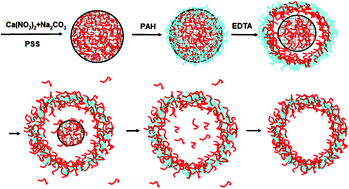
Complex hollow microcapsules were fabricated by adsorbing polyelectrolytes (PEs) onto CaCO3 particles doped with oppositely charged PEs, followed by dissolution of the CaCO3 particles with disodium ethylene diamine tetraacetate dihydrate (EDTA). The shells were formed in situ by complexation of the oppositely charged PEs they encountered during core removal. For this context, sodium poly(styrene sulfonate) (PSS) and poly(allylamine hydrochloride) (PAH) are exemplified as the doping and adsorbing PEs, respectively. Characterization by modern techniques confirms the hollow nature and integrity of the resulting microcapsules. The capsules expanded their size to 150% of their templates due to the osmotic tension and plastic deformation of the primary shells. A microcapsule with a diameter of 7.5 µm weighed 3.8 pg, in which 74 wt% was contributed by PSS and 26 wt% by PAH. This created a negatively charged surface of the microcapsule (−33 ± 8.3 mV). Calculation revealed further that 75% of the initially doped PSS was released. An osmotic induced invagination method detected an elasticity modulus of 140 MPa for the complex PAH–PSS shell, which is only half of the value (290 MPa) of the orderly assembled PAH–PSS multilayers. This new technique has the advantages of time-saving and materials-saving, and can be easily scaled-up.

 Please wait while we load your content...
Please wait while we load your content...