18-Membered cyclic esters derived from glycolide and lactide: preparations, structures and coordination to sodium ions†‡
Abstract
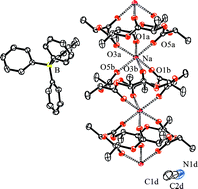
From reactions between glycolide or lactide (4 equiv.) with 4-dimethylaminopyridine, DMAP (1 equiv.) and NaBPh4 (1 equiv.) in benzene at 70 °C the cyclic ester adducts (CH2C(O)O)6NaBPh4 and (CHMeC(O)O)6NaBPh4 are formed respectively. The structures of the salts Na[(S,R,S,R,S,R)-(CH3CHC(O)O)6]2BPh4·CH3CN and (CH2C(O)O)6NaBPh4·(CH3CN)2 are reported. The cyclic esters were separated by chromatography and the structures of (CH2C(O)O)6, (S,R,R,R,R,R)-(CHMeC(O)O)6 and (S,S,R,R,R,R)-(CHMeC(O)O)6 were determined. The 1H and 13C NMR data are reported for one of each of the six enantiomers of (CHMeC(O)O)6 and the two meso isomers. The mechanism for the formation of these 18-membered rings is discussed in terms of an initial reaction between DMAP and NaBPh4 in hot benzene that produces NaPh and DMAP : BPh3 in the presence of the monomer lactide. The cyclic esters (CHMeC(O)O)6 can also be obtained from the reaction between polylactide, PLA, in the presence of DMAP and NaBPh4. The cyclic esters 3-methyl-1,4-dioxane-2,5-dione and 3,6,6-trimethyl-1,4-dioxane-2,5-dione undergo similar ring enlarging reactions to give cyclic 18-membered ring esters as determined by ESI-MS.

 Please wait while we load your content...
Please wait while we load your content...