Physical properties of soft repulsive particle fluids
Abstract
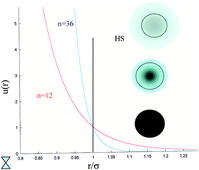
Molecular dynamics computer simulation has been applied to inverse power or soft-sphere fluids, in which the particles interact through the soft-sphere pair potential, ϕ(r) = ε(σ/r)n, where n measures the steepness or stiffness of the potential, and ε and σ are a characteristic energy and distance, respectively. The focus of the study is on very soft particles with n values down to 4 considered, at densities up to and along the fluid–solid co-existence density. It is shown that in the soft-particle limit the local structure is dominated by the lengthscale associated with the average nearest neighbour distance of a random structure, which is ∝ρ−1/3 and increasingly only very weakly dependent on n. This scaling is also manifest in the behaviour of the average energy per particle with density. The self-diffusion coefficient and shear viscosity are computed along the fluid–solid co-existence line as a function of n, for the first time. The product Dηs steadily increases with softness for n < 10, whereas the modified Stokes–Einstein relationship of Zwanzig, Dηs/ρ1/3, where ρ is the number density, is within statistics constant over the same softness range. This is consistent with our observation that the static properties are determined by a characteristic lengthscale (i.e., l) which is ∝ρ−1/3 in the soft-particle limit. The high frequency elastic moduli of these fluids are examined, which reveals that the mechanical properties become more ‘rubbery’ as the particles get softer.

 Please wait while we load your content...
Please wait while we load your content...