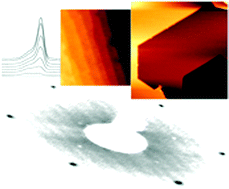
Two different approaches under ambient conditions were developed for the preparation of clean, non-reconstructed, single crystalline ZnO(0001)–Zn surfaces. The surface preparation by a wet chemical etching procedure was compared with the same treatment in combination with a subsequent heat treatment in humidified oxygen atmosphere. Depending on the preparation technique, atomically flat terraces with a width of 100 nm to several micrometers were observed using an atomic force microscope (AFM). The obtained surface structures were further characterized by means of angle resolved X-ray photoelectron spectroscopy (AR-XPS), time-of-flight secondary ion mass spectroscopy (ToF-SIMS), Auger electron spectroscopy (AES) and low energy electron diffraction (LEED) measurements. Based on these results it is shown that the obtained surfaces are, in contrast to surfaces prepared under UHV conditions, stabilised by the adsorption of a monolayer of hydroxides. The important role of H2O during the heat treatment is pointed out by comparing the results of the same heat treatment in the absence of water. H2O turned out to play an important role in the reorganization process of the surface at elevated temperatures, thereby yielding extremely large atomically flat terraces. The terminating edges of these terraces were found to include 120° and 60° angles, thus perfectly reflecting the hexagonal surface structure.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...