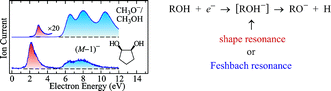
We studied dissociative electron attachment to a series of compounds with one or two hydroxyl groups. For the monoalcohols we found, apart from the known fragmentations in the 6–12 eV range proceeding via Feshbach resonances, also new weaker processes at lower energies, around 3 eV. They have a steep onset at the dissociation threshold and show a dramatic D/H isotope effect. We assigned them as proceeding via shape resonances with temporary occupation of σ*O–H orbitals. These low energy fragmentations become much stronger in the larger molecules and the strongest DEA process in the compounds with two hydroxyl groups, which thus represent an intermediate case between the behavior of small alcohols and the sugar ribose which was discovered to have strong DEA fragmentations near zero electron energy [S. Ptasińska, S. Denifl, P. Scheier and T. D. Märk, J. Chem. Phys., 2004, 120, 8505]. Above 6 eV, in the Feshbach resonance regime, the dominant process is a fast loss of a hydrogen atom from the hydroxyl group. In some cases the resulting (M – 1)– anion (loss of hydrogen atom) is sufficiently energy-rich to further dissociate by loss of stable, closed shell molecules like H2 or ethene. The fast primary process is state- and site selective in several cases, the negative ion states with a hole in the nO orbital losing the OH hydrogen, those with a hole in the σC–H orbitals the alkyl hydrogen.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...