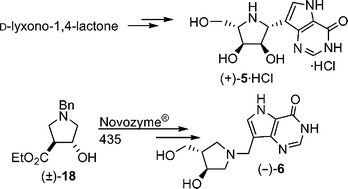
(1R)-1-(9-Deazahypoxanthin-9-yl)-1,4-dideoxy-1,4-imino-L-ribitol [(+)-5] and (3S,4S)-1-[(9-deazahypoxanthin-9-yl)methyl]-4-(hydroxymethyl)pyrrolidin-3-ol [(−)-6] are the L-enantiomers of immucillin-H (D-ImmH) and DADMe-immucillin-H (D-DADMe–ImmH), respectively, these D-isomers being high affinity transition state analogue inhibitors of purine nucleoside phosphorylases (PNPases) developed as potential pharmaceuticals against diseases involving irregular activation of T-cells. The C-nucleoside hydrochloride D-ImmH [(−)-5)·HCl], now “Fodosine™” is in phase II clinical trials as an anti-T-cell leukaemia agent, while D-DADMe–ImmH is a second generation inhibitor with extreme binding to the target enzyme and has entered the clinic for phase I testing as an anti-psoriasis drug. Since the enantiomers of some pharmaceuticals have revealed surprising biological activities, the L-nucleoside analogues (+)-5·HCl and (−)-6, respectively, of D-ImmH and D-DADMe–ImmH, were prepared and their PNPase binding properties were studied. For the synthesis of compound (−)-6 suitable enzyme-based routes to the enantiomerically pure starting material (3S,4S)-4-(hydroxymethyl)pyrrolidin-3-ol [(−)-16] and its enantiomer were developed. The L-enantiomers (+)-5·HCl and (−)-6 bind to the PNPases approximately 5- to 600-times less well than do the D-compounds, but nevertheless remain powerful inhibitors with nanomolar dissociation constants.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...