The effect of light and iron(ii)/iron(iii) on the distribution of Tl(i)/Tl(iii) in fresh water systems†
Abstract
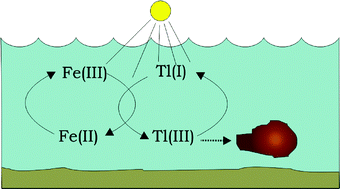
The distribution of aqueous Tl(I)/Tl(III) as a function of light exposure and solution properties was studied by quantifying the oxidation states after separation with ion chromatography and on-line detection with ICP-MS. Ultraviolet irradiation of aqueous solutions containing 1 μg l−1 Tl(III) and in equilibrium with the atmosphere increases the reduction rate. In systems with photoreduction of Fe(III)(aq) a quantitative oxidation of Tl(I)(aq) was observed, notably at low pH. The process is reversible, as indicated by formation of Tl(I) when the irradiated systems were kept in the dark. In systems with colloidal silica-stabilised ferrihydrite, UV irradiation also leads to oxidation of Tl(I)(aq), but not quantitatively. It is suggested that adsorption of thallium to the ferrihydrite determines the rate of oxidation. Detectable, but not quantitative, oxidation of Tl(I)(aq) took place when natural water samples with 1 μg l−1 Tl(I)(aq) were exposed to either sunlight or UV-light. For these samples, the reduction was not quantitative when they were kept in the dark for 24 h. The results suggest that the light dependent iron cycle in fresh water systems strongly influences the redox state of thallium.

 Please wait while we load your content...
Please wait while we load your content...