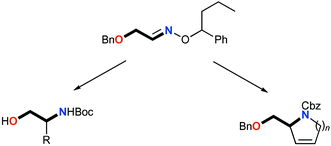
Addition of a range of organolithium and Grignard reagents to (E)-O-(1-phenylbutyl)benzyloxyacetaldoxime 1 in the presence of boron trifluoride diethyl etherate is highly diastereoselective. The resulting hydroxylamines 2 undergo N–O bond cleavage upon treatment with zinc–acetic acid or molybdenum hexacarbonyl to give, after N-protection, protected 1,2-aminoalcohols 3 in high enantiomeric purity. Debenzylation of 3a and 3d gave N-Boc (R)-alaninol and (S)-phenylalaninol respectively. The hydroxylamines 2 also serve as α-amino acid precursors, 2i being converted into N-formyl-(R)-alaninyl-(S)-(4-bromo)phenylalanine ester 7, the N-terminal dipeptide of a natural depsipeptide. The versatility of the 1,2-aminoalcohol derivatives was further illustrated by their conversion into 5-, 6- and 7-membered 2-hydroxymethyl nitrogen heterocycles 15–19 in high enantiomeric excess by a ring-closing metathesis reaction. Further reaction of the dihydropyrrole 15 gave the iminosugar 1,4-dideoxy-1,4-imino-D-ribitol.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...