Metal-assisted red light-induced DNA cleavage by ternary l-methionine copper(ii) complexes of planar heterocyclic bases†
Abstract
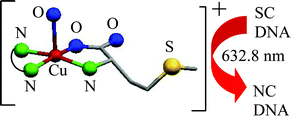
Ternary copper(II) complexes [Cu(L-met)B(Solv)](ClO4) (1–4), where B is a N,N-donor heterocyclic base like 2,2′–bipyridine (bpy, 1), 1,10-phenanthroline (phen, 2), dipyrido[3,2-d:2′,3′-f]quinoxaline (dpq, 3) and dipyrido[3,2-a:2′,3′-c]phenazene (dppz, 4), are prepared and their DNA binding and photo-induced DNA cleavage activity studied (L-Hmet = L-methionine). Complex 2, structurally characterized by X-ray crystallography, shows a square pyramidal (4 + 1) coordination geometry in which the N,O-donor L-methionine and N,N-donor heterocyclic base bind at the basal plane and a solvent molecule is coordinated at the axial site. The complexes display a d–d band at ∼600 nm in DMF and exhibit a cyclic voltammetric response due to the Cu(II)/Cu(I) couple near −0.1 V in DMF–Tris-HCl buffer. The complexes display significant binding propensity to the calf thymus DNA in the order: 4 (dppz) > 3 (dpq) > 2 (phen) ≫ 1 (bpy). Control cleavage experiments using pUC19 supercoiled DNA and distamycin suggest major groove binding for the dppz and minor groove binding for the other complexes. Complexes 2–4 show efficient DNA cleavage activity on UV (365 nm) or red light (632.8 nm) irradiation via a mechanistic pathway involving formation of singlet oxygen as the reactive species. The DNA cleavage activity of the dpq complex 3 is found to be significantly more than its dppz and phen analogues.

 Please wait while we load your content...
Please wait while we load your content...