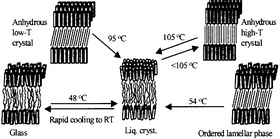
The thermotropic phase behaviour and phase structure of crystalline and non-crystalline n-tetradecyl-β-D-maltoside (C14G2) and n-hexadecyl-β-D-maltoside (C16G2) have been investigated by means of differential scanning calorimetry and X-ray techniques. Upon lyophilisation, both compounds form a solid, lamellar phase comprising disordered head groups and hexagonally packed alkyl chains that are suggested to be tilted and interdigitated. This ordered lamellar phase melts into a metastable lamellar liquid crystal, which re-crystallises to a high-temperature crystalline polymorph comprising interdigitated, non-tilted alkyl chains. Remarkably, the high-temperature polymorph of C14G2 has the same melting point as that of C16G2, namely 105 °C for both surfactants. A low-temperature polymorph of anhydrous C14G2 crystallises from water at room temperature, whereas the hemihydrate of C14G2 crystallises at 6 °C from water, or from chloroform containing trace water. X-ray data suggest both these crystalline modifications to comprise interdigitated and tilted alkyl chains.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...