Synthesis of cycloalkynesvia photochemical decarboxylation of ω-phthalimidoalkynoates†
Abstract
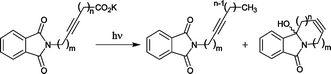
The photodecarboxylation reactions of ω-phthalimidoalkynoates have been studied and the influence of the geometrically restricted acetylene linker on the photocyclization efficiency investigated. Less flexible starting materials give solely CO2H/H-exchange products in good yields. In contrast, derivatives with appropriate chain lengths give the corresponding

 Please wait while we load your content...
Please wait while we load your content...