Linear 1,4-coupled oligoanilines of defined length: preparation and spectroscopic properties
Abstract
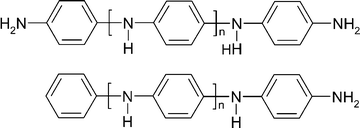
A facile synthetic method involving SNAr coupling of 4-fluoronitrobenzene to arylamines, followed by reduction of the nitro groups, has been developed. The method allows the preparation of symmetric (NH2/NH2 end-capped) and asymmetric (Ph/NH2 end-capped) oligoanilines of even or odd chain lengths. The preparation of the NH2/NH2 end-capped trimer to octamer and Ph/NH2 end-capped trimer to pentamer is described. The effect of the chain length on the spectroscopic properties of the oxidized oligomers has been investigated by UV-Vis-NIR spectroscopy.

 Please wait while we load your content...
Please wait while we load your content...