Peroxo intermediates are implicated in the catalytic cycles of iron enzymes involved in dioxygen metabolism. X-ray absorption spectroscopy has been used to gain insight into the iron coordination environments of the low-spin complex [FeIII(Me-TPEN)(η1-OOH)]2+
(1) and the high-spin complex [FeIII(Me-TPEN)(η2-O2)]+
(2)
(the neutral pentadentate N-donor ligand Me-TPEN =
N-methyl-N,N′,N′-tris(2-pyridylmethyl)ethane-1,2-diamine) and obtain metrical parameters unavailable from X-ray crystallography. The complexes exhibit relatively large pre-edge peak areas of approximately 15 units, indicative of iron centers with significant distortions from centrosymmetry. These distortions result from the binding of peroxide, either end-on hydroperoxo for 1
(rFe–O
= 1.81 Å) or side-on peroxo for 2
(rFe–O
= 1.99 Å). The XAS analyses of 1 strongly support a six-coordinate low-spin iron(III) center coordinated to five nitrogen atoms from Me-TPEN and one oxygen atom from an end-on hydroperoxide ligand. However, the XAS analyses of 2 are not conclusive: Me-TPEN can act either as a pentadentate ligand to form a seven-coordinate peroxo complex, which has precedence in the DFT geometry optimization of [FeIII(N4Py)(η2-O2)]+
(the neutral pentadentate N-donor ligand N4Py =
N,N-bis(2-pyridylmethyl)-N-bis(2-pyridyl)methylamine), or as a tetradentate ligand with a dangling pyridylmethyl arm to form a six-coordinate peroxo complex, which is precedented by the crystal structure of [Fe2III(Me-TPEN)2(Cl)2(μ-O)]2+.
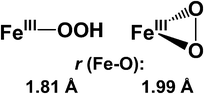
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
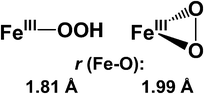

 Please wait while we load your content...
Please wait while we load your content...