Experimental investigations and ab initio studies of selenium(ii) dialkanethiolates, Se(SR)2
Abstract
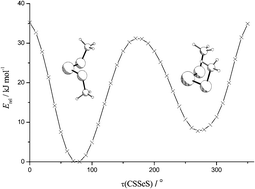
Selenium(II) dimethanethiolate, Se(SMe)2, was synthesized by reaction of SeO2 with HSMe. Basic spectroscopic data for Se(SMe)2 and selenium(II) bis(2-methyl-2-propanethiolate), Se(StBu)2, were recorded and interpreted with the support of ab initio calculations. Both compounds are thermodynamically unstable relatively to selenium and the corresponding disulfide. The UV/vis spectra of both compounds are qualitatively similar, the two bands being attributed to n(Se)–σ*(Se–S) transitions. The bands at 369 and 397 cm−1 in the IR spectra of Se(SMe)2 and Se(StBu)2, respectively, are assigned to νas(SeS2). The 77Se NMR shifts of Se(SMe)2 (784 ppm) and Se(StBu)2 (556 ppm) differ substantially from each other and show positive temperature gradients. Calculations at the GIAO-HF/962+(d) level reproduced the difference of the 77Se NMR chemical shifts between Se(SMe)2 and Se(StBu)2. At the same level, the effect of conformational changes on 77Se shifts were studied for Se(SMe)2. In the solid state Se(SMe)2 forms long intermolecular Se⋯S contacts while Se(StBu)2 does not. Both compounds exhibit anti-conformations of the methyl and tert-butyl groups with respect to the SeS2 plane. MP2/LANL2DZ(d) geometry optimizations, single point energy and frequency calculations performed for Se(SMe)2 show, that syn- (Cs) and anti-conformers (C2) represent minima on the potential energy surface, the latter being by 8 kJ mol−1 lower in energy than the former. Both conformers are stabilized by intramolecular π-type n(S1)–σ*(Se–S2) orbital interactions. The energy of the transition state for the mutual conversion of the two conformers was calculated to be 31 kJ mol−1 above that of the syn conformer, allowing a rapid interconversion of the two conformers at room temperature. Intermolecular interactions between Se(SMe)2 molecules were also studied by means of calculations at the MP2/LANL2DZ(d) level. For Se(StBu)2 MP2/LANL2DZ(d) geometry optimizations and single point energy calculations revealed a C2-symmetric anti- and a C1 symmetric syn-conformer, the latter being 21 kJ mol−1 higher in energy than the former. Se(SMe)2 and Se(StBu)2 exchange thiolate groups with other selenium(II) dithiolates, tellurium(II) dithiolates and with thiols, if catalytic amounts of p-CH3C6H4SO3H are added.

 Please wait while we load your content...
Please wait while we load your content...