Regioselective HON-addition of bifunctional hydrazone oximes to Pt(iv)-bound nitriles†
Abstract
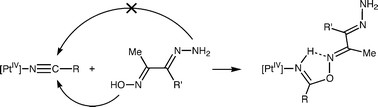
Treatment of trans-[PtCl4(RCN)2]
(R = Me, Et) with the hydrazone oximes MeC(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NOH)C(R′)
NOH)C(R′)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NNH2
(R′
= Me, Ph) at 45 °C in CH2Cl2 led to the formation of trans-[PtCl4{NH
NNH2
(R′
= Me, Ph) at 45 °C in CH2Cl2 led to the formation of trans-[PtCl4{NH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(R)ON
C(R)ON![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(Me)C(R′)
C(Me)C(R′)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NNH2}2]
(R/R′
= Me/Ph 1, Et/Me 2, Et/Ph 3) due to the regioselective OH-addition of the bifunctional MeC(
NNH2}2]
(R/R′
= Me/Ph 1, Et/Me 2, Et/Ph 3) due to the regioselective OH-addition of the bifunctional MeC(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NOH)C(R′)
NOH)C(R′)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NNH2 to the nitrile group. The reaction of 3 and Ph3P
NNH2 to the nitrile group. The reaction of 3 and Ph3P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHCO2Me allows the formation of the Pt(II) complex trans-[PtCl2{NH
CHCO2Me allows the formation of the Pt(II) complex trans-[PtCl2{NH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(Et)ON
C(Et)ON![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(Me)C(Ph)
C(Me)C(Ph)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NNH2}2]
(4). In 4, the imine ligand was liberated by substitution with 2 equivalents of bis(1,2-diphenylphosphino)ethane (dppe) in CDCl3 to give, along with the free ligand, the solid [Pt(dppe)2]Cl2. The free iminoacyl hydrazone, having a restricted life-time, decomposes at 20–25 °C in about 20 h to the parent organonitrile and the hydrazone oxime. The Schiff condensation of the free NH2 groups of 4 with aromatic aldehydes, i.e. 2-OH-5-NO2-benzaldehyde and 4-NO2-benzaldehyde, brings about the formation of the platinum(II) complexes trans-[PtCl2{NH
NNH2}2]
(4). In 4, the imine ligand was liberated by substitution with 2 equivalents of bis(1,2-diphenylphosphino)ethane (dppe) in CDCl3 to give, along with the free ligand, the solid [Pt(dppe)2]Cl2. The free iminoacyl hydrazone, having a restricted life-time, decomposes at 20–25 °C in about 20 h to the parent organonitrile and the hydrazone oxime. The Schiff condensation of the free NH2 groups of 4 with aromatic aldehydes, i.e. 2-OH-5-NO2-benzaldehyde and 4-NO2-benzaldehyde, brings about the formation of the platinum(II) complexes trans-[PtCl2{NH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(Et)ON
C(Et)ON![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(Me)C(Ph)
C(Me)C(Ph)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NN
NN![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH(C6H3-2-OH-5-NO2}2]
(5) and trans-[PtCl2{NH
CH(C6H3-2-OH-5-NO2}2]
(5) and trans-[PtCl2{NH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(Et)ON
C(Et)ON![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(Me)C(Ph)
C(Me)C(Ph)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NN
NN![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH(C6H4-4-NO2}2]
(6), respectively, containing functionalized remote peripherical groups. Metallization of 5, which can be considered as a novel type of metallaligand, was achieved by its reaction with M(OAc)2·nH2O (M = Cu, n
= 2; M = Co, n
= 4) in a 1 ∶ 1 molar ratio furnishing solid heteronuclear compounds with composition [Pt]
∶
[M]
= 1 ∶ 1. The complexes were characterized by C, H, N elemental analyses, FAB+ mass-spectrometry, IR, 1H, 13C{1H} and 195Pt NMR spectroscopies; X-ray structures were determined for 3, 4 and 5.
CH(C6H4-4-NO2}2]
(6), respectively, containing functionalized remote peripherical groups. Metallization of 5, which can be considered as a novel type of metallaligand, was achieved by its reaction with M(OAc)2·nH2O (M = Cu, n
= 2; M = Co, n
= 4) in a 1 ∶ 1 molar ratio furnishing solid heteronuclear compounds with composition [Pt]
∶
[M]
= 1 ∶ 1. The complexes were characterized by C, H, N elemental analyses, FAB+ mass-spectrometry, IR, 1H, 13C{1H} and 195Pt NMR spectroscopies; X-ray structures were determined for 3, 4 and 5.

 Please wait while we load your content...
Please wait while we load your content...