Alkene metatheses in transition metal coordination spheres: dimacrocyclizations that join trans positions of square-planar platinum complexes to give topologically novel diphosphine ligands
Abstract
The alkene-containing ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2)2)2
(4) are prepared from PPhH2, n-BuLi, and the corresponding
CH2)2)2
(4) are prepared from PPhH2, n-BuLi, and the corresponding ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2)2)2
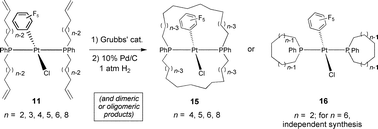
(11, 93–73%; n
=
a, 2; b, 3; c, 4; d, 5; e, 6; f, 8). Ring-closing metatheses with Grubbs' catalyst (2) are studied. With 11e, two isomers of trans-(Cl)(C6F5)
CH2)2)2
(11, 93–73%; n
=
a, 2; b, 3; c, 4; d, 5; e, 6; f, 8). Ring-closing metatheses with Grubbs' catalyst (2) are studied. With 11e, two isomers of trans-(Cl)(C6F5)![[lower bond 1 start]](https://www.rsc.org/images/entities/char_e012.gif) Pt(
Pt(![[upper bond 1 start]](https://www.rsc.org/images/entities/char_e010.gif) PPh(CH2)14P
PPh(CH2)14P![[lower bond 1 end]](https://www.rsc.org/images/entities/char_e013.gif) (C
(C![[upper bond 1 end]](https://www.rsc.org/images/entities/char_e011.gif) H2)14Ph)
(15e) are isolated after hydrogenation. Both form via dimacrocyclization between the trans-phosphine
H2)14Ph)
(15e) are isolated after hydrogenation. Both form via dimacrocyclization between the trans-phosphine ![[upper bond 1 start]](https://www.rsc.org/images/entities/char_e010.gif) PPh(C
PPh(C![[upper bond 1 end]](https://www.rsc.org/images/entities/char_e011.gif) H2)14)2
(16e) is independently prepared by (i) protecting 4e as a borane adduct, H3B·PPh((CH2)6CH
H2)14)2
(16e) is independently prepared by (i) protecting 4e as a borane adduct, H3B·PPh((CH2)6CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2)2, (ii) cyclization with 2 and hydrogenation to give H3B·
CH2)2, (ii) cyclization with 2 and hydrogenation to give H3B·![[upper bond 1 start]](https://www.rsc.org/images/entities/char_e010.gif) PPh(C
PPh(C![[upper bond 1 end]](https://www.rsc.org/images/entities/char_e011.gif) H2)14, (iii) deprotection and reaction with 12. The sample derived from 11e contains ≤2%
16e;
H2)14, (iii) deprotection and reaction with 12. The sample derived from 11e contains ≤2%
16e; ![[upper bond 1 start]](https://www.rsc.org/images/entities/char_e010.gif) PPh(CH2)2CH
PPh(CH2)2CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH(C
CH(C![[upper bond 1 end]](https://www.rsc.org/images/entities/char_e011.gif) H2)2)2
(86%), as confirmed by a crystal structure of the hydrogenation product. Although 11b does not yield tractable products, 11c gives syn-(E,E)-trans-(Cl)(C6F5)
H2)2)2
(86%), as confirmed by a crystal structure of the hydrogenation product. Although 11b does not yield tractable products, 11c gives syn-(E,E)-trans-(Cl)(C6F5)![[lower bond 1 start]](https://www.rsc.org/images/entities/char_e012.gif) Pt(
Pt(![[upper bond 1 start]](https://www.rsc.org/images/entities/char_e010.gif) PPh(CH2)4CH
PPh(CH2)4CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH(CH2)4P
CH(CH2)4P![[lower bond 1 end]](https://www.rsc.org/images/entities/char_e013.gif) (CH2)4CH
(CH2)4CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH(C
CH(C![[upper bond 1 end]](https://www.rsc.org/images/entities/char_e011.gif) H2)4Ph)
(21%). This structure, and that of the hydrogenation product (syn-15c; 95%), are verified crystallographically. Analogous sequences with 11d,f give syn-15d,f
(5 and 14% overall).
H2)4Ph)
(21%). This structure, and that of the hydrogenation product (syn-15c; 95%), are verified crystallographically. Analogous sequences with 11d,f give syn-15d,f
(5 and 14% overall).

 Please wait while we load your content...
Please wait while we load your content...