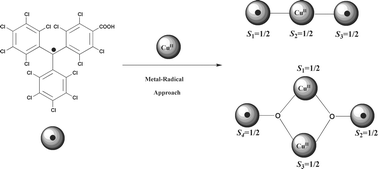
A series of complexes of copper(II)-containing a perchlorotriphenylmethyl radical functionalized with a carboxylic group as a new ligand is reported. The compounds [Cu(PTMMC)2(L)3]
(PTMMC = (tetradecachloro-4-carboxytriphenyl)methyl radical; L =
(1) H2O, (2) pyrimidine and ethanol or (3) pyridine), [Cu2(PTMMC)2(MeCOO)2(H2O)2]
(4) and [Cu(HPTMMC)2(L)3]
(HPTMMC =
α-H-(tetradecachlorotriphenyl)methane-4-carboxylic acid; L = pyridine)
(5) were structurally characterized. In complexes 1, 2, 3 and 5, the copper(II) ion is coordinated to two PTMMC (or HPTMMC) units in a slightly distorted square planar surrounding, while 4 shows a paddle-wheel copper(II) dimer structure, where each Cu metal ion has four O atoms of different carboxylate groups, two of them belonging to two PTMMC radicals. The copper(II)–radical exchange couplings are antiferromagnetic for complexes 1, 2 and 3. A linear three-spin model was applied to complexes 1, 2 and 3 to give J/kB
=
−24.9, −15.0 and −20.7 K, respectively. Magnetic properties of 4 show that it is one of the scarce examples of a spin-frustrated system composed of organic radicals and metal ions. In this case, experimental data were fitted to a magnetic model based on a symmetrical butterfly arrangement to give a copper(II)–copper(II) exchange coupling of J/kB
=
−350.0 K and a copper(II)–radical exchange coupling of J/kB
=
−21.3 K, similar to that observed for the copper(II)–radical interactions in complexes 1, 2 and 3.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...