Zwitterionic macrocyclic metal sulfate extractants containing 3-dialkylaminomethylsalicylaldimine units
Abstract
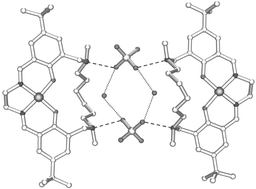
10,25-Di-tert-alkyl-14,21-dimethyl-3,6,14,21-tetraazatricyclo[21.3.1.18,12]octacosa-1(27),2,6,8,10,12(28),23,25-octaene-27,28-diol macrocycles form Ni(II) and Cu(II) complexes in which the metal cation and the sulfate anion are bound in separated sites in a zwitterionic form of the ligand. The nonyl-substituted macrocycle shows a higher affinity for SO42− and a lower binding strength for Cu2+ than open chain analogues, the pH-dependences for which fall in ranges which allow loading of CuSO4 at pH ≈ 4 and easy stripping to recycle the ligand. X-Ray structure determinations of the Cu(II) and Ni(II) sulfate complexes of the tert-butyl substituted ligand suggest that the de-tuning of M2+-binding results from a distortion from planarity of the “salen” N2O22− donor set imposed by the incorporation of the hexamethylene strap in the ligand and reveal that the sulfate is bound as a hydrate in a 2 ∶ 2 ∶ 2 ∶ 2, ligand–M2+–SO42−–H2O, assembly.

 Please wait while we load your content...
Please wait while we load your content...