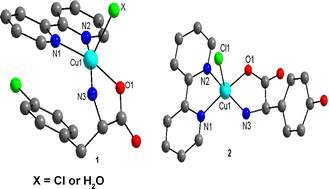
The mixed ligand complexes {[Cu(4Cl-Phe)(bpy)(H2O)]Cl·[Cu(4Cl-Phe)(bpy)Cl]·2H2O}
(1) and [Cu(4HPG)(bpy)]·2H2O (2) were synthesized and their crystal structures determined by X-ray crystallography (4Cl-Phe =
D,L-4-chlorophenylalaninato, 4HPG =
D-4-hydroxyphenylglycinato, bpy = 2,2′-bipyridine). The asymmetric unit of crystals of 1 consists of two slightly different Cu(II) complexes-one contains an axial water ligand, whereas the second one has a chloride anion at that position. By contrast, 2 exhibits two close similar molecules, both having axial chloride ligands, and non coordinated water. The geometrical parameters for π,π-stacking interactions were determined by an application of PLATON program. Intermolecular π,π-interaction between bpy ligands on adjacent Cu(II) moieties results in homo pairs of complexes in the case of 1. In addition, both species of compound 1 exhibit intramolecular interligand π,π-interaction between the 4Cl-phenyl ring of 4Cl-Phe and the metalloaromatic Cu–bpy chelate ring. In crystal 2, multi-stacked homo chains are formed. Analogous calculations for previously reported, related compounds, similar to those presented here, revealed that in most cases with intra-molecular interligand π,π-interaction there is an efficient “aryl–metal chelate ring”
π,π-stacking instead of an aryl–aryl interligand π,π-interaction, which indeed is not observed. These findings let us to take a critical look at the nature of the intramolecular interligand π,π-stacking interaction in the referred copper(II) mixed-ligand complexes.

 Please wait while we load your content...
Please wait while we load your content...