Synthesis of sparteine-like chiral diamines and evaluation in the enantioselective lithiation–substitution of N-(tert-butoxycarbonyl)pyrrolidine†
Abstract
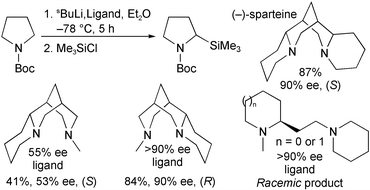
Three chiral diamines were synthesised and evaluated as sparteine surrogates in the lithiation–substitution of N-(tert-butoxycarbonyl)pyrrolidine. The synthesis and attempted resolution of sparteine-like diamines {(1S*,2R*,8R*)-10-methyl-6,10-diazatricyclo[6.3.1.02,6]dodecane and (1S*,2R*,9R*)-11-methyl-7,11-diazatricyclo[7.3.1.02,7]tridecane} (via inclusion complex formation) are reported. Unfortunately, it was only possible to resolve the diazatricyclo[7.3.1.02,7]tridecane compound. An alternative route to (1R,2S,9S)-11-methyl-7,11-diazatricyclo[7.3.1.02,7]tridecane starting from the natural product, (−)-cytisine, is described. This simple three-step route furnished gram-quantities of a (+)-sparteine surrogate. X-Ray crystallography of an intermediate in the route, (1R,5S,12S)-3-methoxycarbonyldecahydro-1,5-methanopyrido[1,2-a][1,5]diazocin-8-one, enabled the stereochemistry of all of the tricyclic diamines described in this paper to be unequivocally established. Two other diamines, starting from (S)-proline and resolved 2-piperidine ethanol, were prepared using standard methods. These diamines lacked the bispidine framework of (−)-sparteine and were found to impart vastly inferior enantioselectivity. It was concluded that, for the asymmetric lithiation–substitution of N-Boc pyrrolidine, a rigid bispidine framework and only three of the four rings of (−)-sparteine are needed for high enantioselectivity. Furthermore, it is shown that diamine (1R,2S,9S)-11-methyl-7,11-diazatricyclo[7.3.1.02,7]tridecane is the first successful (+)-sparteine surrogate.

 Please wait while we load your content...
Please wait while we load your content...