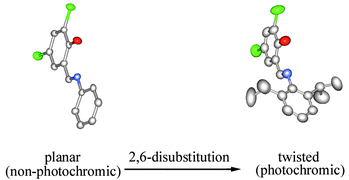
N-(3,5-Dihalosalicylidene)-2,6-dialkylaniline derivatives were prepared and their structure and photochromic properties were investigated. From the X-ray crystallography, it was revealed that steric repulsion between the azomethine hydrogen atom and the alkyl groups at the 2,6-positions of the aniline ring lead to a non-planar molecular structure, which was effective for the crystals to exhibit photochromism. The relationship between photochromicity and crystal packing of N-(3,5-dichlorosalicylidene)-2,6-dialkylanilines series was also discussed. The 2,6-dialkylaniline method was suggested to be applicable to the preparation of photochromic Schiff bases with various substituents in the salicylidene rings.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...