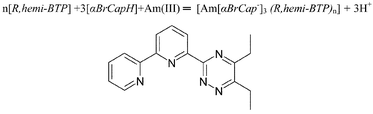
It has been established that 6-(5,6-dialkyl-1,2,4-triazin-3-yl)-2,2′-bipyridines (R,hemi-BTPs) have properties which are intermediate between those of the terpyridines and the bis(1,2,4-triazin-3-yl)pyridines (BTPs). However, they resemble the terpyridines much more closely than the BTPs. It has been shown that Et,hemi-BTP when dissolved in TPH—a dodecane-like solvent—is a selective reagent for the separation of americium(III) from europium(III). Solution NMR in acetonitrile largely confirmed the crystallographic results. There was no evidence for a 1 ∶ 3 complex cation, or for significant differences between metal(III)–N distances for the pyridine and 1,2,4-triazine rings. Intramolecular hydrogen bonding plays a crucial role in the formation of metal coordination spheres, which explains the differences between the terpyridyl, R,hemi-BTPs and the BTPs. Protonation of the R,hemi-BTPs facilitates a conformational change which is necessary for complexation.

 Please wait while we load your content...
Please wait while we load your content...