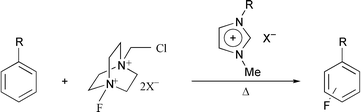
The NF fluorinating agent F-TEDA-BF4 dication salt (Selectfluor™) 1 dissolves in imidazolium-based ionic liquids [emim][OTf] 7, [emim][BF4] 8, [bmim][PF6] 9 and [bmim][BF4] 10 (assisted by sonication), providing a convenient medium for fluorination of arenes under essentially acid-free conditions in a simple set-up (no volatile solvent; simple extraction of the aromatics without aqueous work-up), from which the ionic liquid can be easily recycled and reused. Comparative studies in [emim][OTf] 7 with anisole as substrate show that 1 is superior to NFTh-BF4 (Accufluor®) 2
and that the N-fluoropyridinium salt NFPy-B2F74 is least effective. The scope of the reaction has been surveyed. Substrate selectivity (kmesitylene
∶
kdurene
= 10) measured in competitive experiments in 7 is clearly indicative of a conventional polar mechanism. Substrate selectivity measured without the ionic liquid in MeCN solvent is also indicative of a polar mechanism but exhibits lower magnitude (kmesitylene
∶
kdurene
= 6).
Addition of dicyclohexano-24-crown-8 to the fluorination reaction mixture (1 and anisole) in 7 reduced the conversion but did not change the isomer distribution. AM1 minimization was used to model the complexation of 1
with this crown.
With reactive aromatics optimal fluorination yields in ionic liquids (using 1 equivalent of the NF reagent) are around 50% (higher for naphthalene). A key control experiment suggests that the free base (produced upon transfer-fluorination) could complex to unreacted 1 (generating a bulky dimer complex which may be ineffective for fluorine transfer) in competition to N-protonation.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...