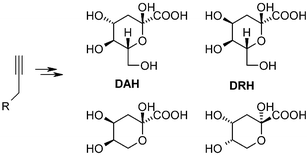
A straightforward synthesis of DAH (3-deoxy-D-arabino-hept-2-ulosonic acid) and DRH (3-deoxy-D-ribo-hept-2-ulosonic acid)
Abstract
Based on the oxidation of corresponding terminal alkynes to α-keto esters, DAH (3-deoxy-D-arabino-hept-2-ulosonic acid) in its pyranose form and DRH (3-deoxy-D-ribo-hept-2-ulosonic acid) in its protected pyranose–furanose mixture form were efficiently synthesized. These terminal alkynes were in turn obtained by C1- or C3-homologation of corresponding protected sugars, respectively. Along with the application of this strategy, the syntheses of two C6-ulosonic acids, 3-deoxy-D-erythro-hex-2-ulosonic acid and 3-deoxy-L-erythro-hex-2-ulosonic acid in their protected pyranose form, were also described.

 Please wait while we load your content...
Please wait while we load your content...