Towards complete stereochemical control: complementary methods for the synthesis of six diastereoisomeric monosaccharide mimetics
Abstract
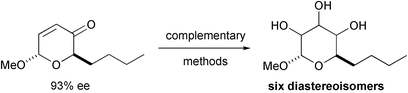
Complementary methods for the synthesis of diastereomeric monosaccharide mimetics are described which rely on the functionalisation of derivatives of 2-butyl-6-methoxy-2,6-dihydropyran-3-one. The stereochemical outcome of dihydroxylation of these derivatives under Upjohn's and Donohoe's (directed) reaction conditions are described. The hydrolyses of diastereomeric 2-butyl-4,5-epoxy-3-hydroxy-6-methoxytetrahydropyrans, generated either by epoxidation with perbenzimidic acid or by cyclisation of the corresponding hydroxy iodides, are described in detail. These methods have enabled stereoselective syntheses of six of a possible eight (ignoring anomers) diastereoisomers of 3,4,5-triacetoxy-2-butyl-6-methoxytetrahydropyran. The power of the general approach lies in the ability to choose at a late stage in the synthesis which diastereoisomer is prepared.

 Please wait while we load your content...
Please wait while we load your content...