AsX3 (X = Cl, Br or I) reacts with one molar equivalent of L [L =
o-C6H4(PMe2)2, o-C6H4(PPh2)2, o-C6H4(AsMe2)2 or MeC(CH2AsMe2)3] in rigorously anhydrous CH2Cl2 or thf solution to give colourless or yellow solids of composition [AsX3L]. Using two molar equivalents of PMe3 with AsX3 affords solids of empirical formula [AsX3(PMe3)2], whereas one molar equivalent of PMe3 with AsCl3 gives the 1 ∶ 1 complex [AsCl3(PMe3)]. Reaction of AsX3 with AsMe3 gives only the 1 ∶ 1 product [AsX3(AsMe3)], whether using a 1 ∶ 1 or 1 ∶ 2 AsCl3
∶ AsMe3 ratio.
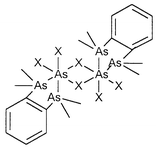
The products have been characterised by microanalysis, and IR, 1H NMR and 31P{1H} NMR spectroscopies, where appropriate. The crystal structures of three species, [AsCl3(PMe3)], [AsBr3{o-C6H4(AsMe2)2}] and [AsI3{o-C6H4(AsMe2)2}] have been determined. [AsCl3(PMe3)] adopts a dimeric structure formed through μ-dichloro-bridged edge-shared square pyramidal moieties with anti PMe3 ligands in the axial sites. Interestingly, in the lattice, there are two crystallographically independent dimers which are arranged orthogonally with long range As⋯Cl contacts between between them, giving rise to infinite chains. Thus, in one dimer the As centres are formally 5-coordinate, while in the adjacent dimer the coordination is [5 + 1]. The complexes
[AsX3{o-C6H4(AsMe2)2}] (X = Br or I) are both dimeric through μ-dihalo-bridged bioctahedra in the isomer (A) form. Trends in bond length and angle distributions are considered and the stereochemical activity of the lone pair on the As(III) acceptor discussed.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...