Activation of H2 by halocarbonyl bis-phosphine and bis-arsine iridium(i) complexes. The use of parahydrogen induced polarisation to detect species present at low concentration and investigate their reactivity†
Abstract
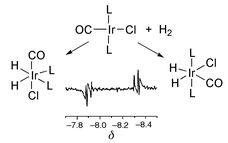
The iridium phosphine complexes Ir(CO)Cl(L)2 [L = PPh3, PMe3, AsPh3 and PPh2Cl, and L2 = (PPh2Cl)(PPh3)] add H2 to form the corresponding dihydrides IrH2(CO)Cl(L)2. These products are detected at enhanced levels of sensitivity through the 1H NMR signatures of their hydride resonances via para-hydrogen (p-H2) based spin state synthesis. Products corresponding to addition across both the Cl–Ir–CO and L–Ir–L axes are detected. For L = PPh3, there is a 100 fold preference for the former pathway at 295 K, while for L = AsPh3 the second product is favoured by a factor of 2.85. At elevated temperatures a third product corresponding to addition over the Cl–Ir–L axis is detected for L = AsPh3 and PPh2Cl. Under these conditions, the CO and HCl transfer products Ir(H)3(CO)2(AsPh3), and IrH(CO)Cl2(AsPh3)2 are also formed in a thermal reaction. When IrH2(CO)Cl(L)2 is warmed or photolysed with H2 and CO, the corresponding products are produced for L = PPh3 and PMe3. However after photolysis with H2 alone Ir(H)3(CO)(L)2 is the favoured product. Additional products detected during the photochemical studies include Ir(H)2(PPh3)(PPh2C5H4CO), an unusual orthometallation product containing an η2-acyl ligand, and the binuclear products H(Cl)Ir(PMe3)2(μ-H)(μ-Cl)Ir(PMe3)(CO) and (H)2Ir(PMe3)2(μ-Cl)2Ir(PMe3)(CO).

 Please wait while we load your content...
Please wait while we load your content...