Efficient one-step synthesis of trialkylsubstituted 2(5H)-furanones utilizing direct Ti-crossed aldol condensation and its application to the straightforward synthesis of (R)-mintlactone and (R)-menthofuran†
Abstract
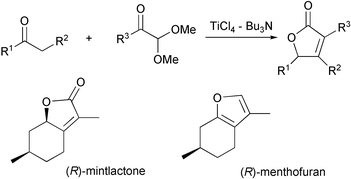
TiCl4–Bu3N-mediated condensation of

 Please wait while we load your content...
Please wait while we load your content...