A series of polyrotaxanes containing α-cyclodextrin and naphthalene-modified α-cyclodextrin and solvent effects on the fluorescence quenching by terminal units
Abstract
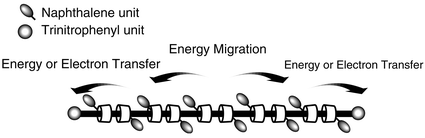
Photoquenching systems were constructed using polyrotaxanes in which cyclodextrin (CD) rings of α-CD and 6-deoxy-6-(-naphthalene-2-sulfonyl)-α-cyclodextrin (NpCD) are threaded by a poly(ethylene glycol) chain with trinitrophenyl (acceptor) units at both ends (8–12). The effects of the component ratio of the polyrotaxanes on the naphthalene fluorescence quenching efficiency and energy migration between naphthalene units were examined by steady-state and time-resolved spectra and anisotropy measurements. The quenching efficiency of 8, which has the lowest content of NpCD, is larger than those of the polyrotaxanes with higher NpCD contents; however this tendency depends on the solvents. We explain the quenching mechanism of naphthalene fluorescence by the combination process of Förster type energy transfer and electron transfer to the trinitrophenyl units.

 Please wait while we load your content...
Please wait while we load your content...