Enantiospecific syntheses of pseudopterosin aglycones. Part 2. Synthesis of pseudopterosin K–L aglycone and pseudopterosin A–F aglycone via a B→BA→BAC annulation strategy
Abstract
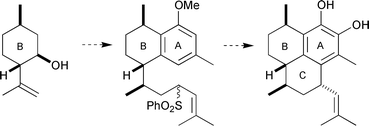
The enantiomeric aglycones of pseudopterosins K–L and A–F are synthesised from (−)- and (+)-isopulegol respectively. Key features are (a) the construction of the C3 stereogenic centre by a directed epoxidation–reduction sequence (K–L); (b) the creation of the C3 stereogenic centre by a Pfaltz asymmetric conjugate reduction (A–F); (c) benzannulation of a cyclic ketone starting with an α-oxoketene-S,S-acetal to give a tetrahydronaphthol ether; and (d) a diastereoselective intramolecular electrophilic aromatic substitution using an allylic sulfone as the electrophilic trigger to complete the hexahydro-1H-phenalene core. An X-ray structure of compound 50 was determined.

 Please wait while we load your content...
Please wait while we load your content...