Abstract
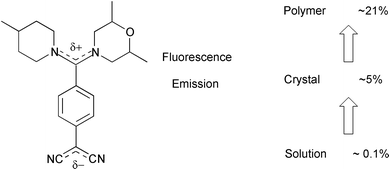
The reactions of primary and secondary amines with 7,7,8,8-tetracyano-p-quinodimethane (TCNQ) lead to mono- and di-substituted adducts. Fluorescence emission has been observed for several of these compounds. The luminescence property of the TCNQ adducts, 2-{4-[(2,6-dimethylmorpholin-4-yl)(4-methylpiperidin-1-yl)methylene]cyclohexa-2,5-dien-1-ylidene}malononitrile (MORPIP) and 2-{4-[cyclohex-1-yltetrahydropyrimidin-2(1H)-ylidene]cyclohexa-2,5-dien-1-ylidene}malononitrile (AMINO) were investigated in a variety of environments. These included alcohol solutions and crystals at room temperature and glass forming solvents and polymer films as a function of temperature. The fluorescence quantum yields and Stokes' shifts were found to be very sensitive to the matrix. Crystal structure data show that the molecules are non-planar in the ground state. The matrix effect is discussed in terms of the conformational change during photo-excitation and the constraint imposed on this by the matrix.

 Please wait while we load your content...
Please wait while we load your content...