Solar-driven simultaneous production of hypochlorous acid and hydrogen from saline water over RhCrOx-loaded SrTiO3 photocatalyst systems†
Sayuri
Okunaka‡
 *,
Toshio
Nakamura
,
Takeshi
Ikeda
,
Kohei
Tsuruda
and
Hiromasa
Tokudome
*,
Toshio
Nakamura
,
Takeshi
Ikeda
,
Kohei
Tsuruda
and
Hiromasa
Tokudome
 *
*
Research Institute, TOTO LTD., 2-8-1 Honson, Chigasaki, Kanagawa, 253-8577, Japan. E-mail: hiromasa.tokudome@jp.toto.com
First published on 24th November 2023
Abstract
Solar-light-driven production of hydrogen (H2) from water using semiconductor photocatalysts is one of the breakthrough technologies of the sustainable and economical solar energy conversion process. However, the conventional oxidation product in water splitting reactions is mainly oxygen gas with low economic benefit. In this paper, photocatalytic simultaneous production of hypochlorous acid (HClO) as a high-value oxidant along with H2 in saline water under simulated solar light was successfully achieved for the first time using SrTiO3:Al photocatalyst systems. Loading of a co-catalyst was found to be essential to proceed the photocatalytic H2/HClO production reaction, in particular the RhCrOx(0.1 wt%)-loaded SrTiO3:Al photocatalyst (RhCrOx/SrTiO3:Al) showed the highest activity. In the RhCrOx, the Rh species acts as a co-catalyst to promote proton reduction, and CrOx suppresses the decomposition of the produced HClO. The HClO production rates on the RhCrOx/SrTiO3:Al photocatalyst increased with increasing the Cl− substrate concentration, whereas HClO was produced even in the diluted NaCl aqueous solution of ca. 0.001 M. Moreover, RhCrOx/SrTiO3:Al photocatalyst films, in which the photocatalyst particles are fixed onto a glass substrate, were prepared by simple screen printing followed by a drying and calcination process. The RhCrOx/SrTiO3:Al photocatalyst film produced H2/HClO from saline water under simulated solar light. The evolution rate of H2/HClO on the RhCrOx/SrTiO3:Al photocatalyst film was found to be almost the same as that on the corresponding suspended particles thanks to the porous structure enabling an efficient mass transfer.
Introduction
Toward the construction of a sustainable society, solar to chemical energy conversion using semiconductor photocatalysts has received significant recent attention because it can be a straightforward and environmentally friendly way to obtain valuable chemicals, and resolve energy and environmental problems.1–4 In particular, solar to H2 conversion through the photocatalytic water splitting reaction has been promised to obtain clean H2 energy without emitting carbon dioxide by utilizing abundant solar light.1,5–9 To date, various semiconductor photocatalysts that can work efficiently under solar-light irradiation have been developed to proceed the water splitting reaction. For example, aluminum-doped strontium titanate (SrTiO3:Al), which is responsive to ultra-violet (UV) light, can efficiently split water with a solar to H2 conversion efficiency of 0.4/0.65% under natural/simulated solar light irradiation.10 Also, various photocatalyst systems have been developed for scalable solar hydrogen production. Besides conventional particle suspension or electrode systems, the panel/sheet systems, in which semiconductor photocatalyst particles are fixed onto a glass substrate, have been paid much attention to efficiently harvest sunlight with dilute energy.11–19 Solar hydrogen production on a 100 m2-sized panel reactor using a SrTiO3:Al photocatalyst has been recently reported.19 However, oxygen (O2) gas produced in water splitting reactions is generally disposed of because of its low economic benefit.20,21 Therefore, it is necessary to develop more economically attractive ways for solar energy conversion.One of the essential strategies lies in the replacement of O2 evolution with oxidative production of high-value-added chemicals, such as hydrogen peroxide (H2O2) or HClO, which is an important chemical substance with a wide range of industrial applications.20–29 In particular, HClO is a well known inorganic oxidant with excellent microbial and oxidizing properties. It is widely used as a disinfectant, deodorizer, and bleaching agent in various industries.30,31 At present, fifty million tons of HClO and its derivatives are produced by electrochemical processes in the world.21
Thus, HClO production via oxidation of Cl− in water by photogenerated holes in photocatalysts can become a valuable and sustainable reaction, along with H2 generation by photogenerated electrons. In this reaction, HClO is produced via oxidation of Cl− by the photogenerated holes; Cl− is first oxidized to Cl2 (eqn (1)) and then the produced Cl2 disproportionates in the aqueous environment to form HClO (eqn (2)). In addition, the reduction reaction of H+ into H2 is carried out by the photogenerated electrons (eqn (3)).
Oxidation reaction:
| 2Cl− → Cl2 + 2e−, E = +1.48 V vs. RHE | (1) |
| Cl2 + H2O ↔ H+ + HClO + Cl− | (2) |
Reduction reaction:
| 2H+ + 2e− = H2, E = 0.0 V vs. RHE | (3) |
Recently, photoelectrochemical HClO production from aqueous NaCl solution has also been demonstrated with a photoelectrochemical system using a BiVO4/WO3 multilayer photoanode under simulated solar light irradiation.24,26,27 Also, some suspension systems including photocatalyst particles such as Pt- or Pt, MnOx-loaded WO3, and Au-loaded AgCl have demonstrated HClO generation in an aqueous solution of NaCl under simulated solar light.25,28,29 However, it was difficult to produce H2 with these photocatalysts because of the difficulty in the reduction of protons (H+) by the photogenerated electrons owing to their deep conduction band edge (>0 V vs. RHE) or the possibility of the self-reduction of Ag+ for AgCl. Therefore, there is still a demand for photocatalysts and their systems that can produce HClO and H2 simultaneously from an aqueous solution containing Cl− under solar light.
To achieve the simultaneous HClO/H2 production on a photocatalyst, choosing photocatalyst materials which have both the band potentials to reduce H+ (<0 V vs. RHE) and oxidize Cl− (>1.48 V vs. RHE) is needed. For example, SrTiO3 is a well-known photocatalyst to split water efficiently and shows a high H2 evolution ability under UV irradiation, and also has the sufficient band potential for producing HClO via oxidation of Cl− theoretically.10,32,33 Therefore, the properties of SrTiO3 photocatalysts motivated us to employ them in the construction of the photocatalytic HClO/H2 production system under solar light. In this study, we used the SrTiO3:Al photocatalyst as a prototype and examined its HClO/H2 production abilities from an aqueous NaCl solution under simulated solar light. We investigated the effect of co-catalyst loading onto the SrTiO3:Al photocatalyst on the HClO production under solar-light irradiation in detail. Moreover, we fabricated a SrTiO3:Al photocatalyst panel by simple screen-printing followed by calcinations, to achieve simultaneous HClO/H2 production under solar light.
Experimental
Materials
Strontium titanate (SrTiO3), rhodium(III) chloride trihydrate (RhCl3·3H2O), hydrogen hexachloroplatinate(IV) hexahydrate (H2PtCl6·6H2O), manganese(II) nitrate hexahydrate (Mn(NO3)2·6H2O), cobalt(II) nitrate hexahydrate (Co(NO3)2·6H2O), nickel(II) nitrate hexahydrate (Ni(NO3)2·6H2O), copper(II) nitrate trihydrate (Cu(NO3)2·3H2O), and sodium chloride (NaCl) were purchased from FUJIFILM Wako Pure Chemical Corporation, Japan. Strontium chloride (SrCl2) and chromium(III) nitrate enneahydrate (Cr(NO3)3·9H2O) were purchased from Kanto Chemical, Ltd., Japan. Aluminum oxide (Al2O3) and ruthenium trichloride trihydrate (RuCl3·3H2O) were purchased from Sigma-Aldrich. All reagents were used as received, and all the experiments were conducted under ambient conditions without eliminating the moisture from the atmosphere.Preparation of co-catalyst loaded SrTiO3:Al photocatalysts
SrTiO3:Al photocatalyst particles were prepared via the flux method which has been reported previously. SrTiO3, Al2O3 and SrCl2 as a flux were mixed at a molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.02
0.02![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, and calcinated in an alumina crucible at 1150 °C for 10 h in air. The obtained white powders were subsequently separated from the flux by washing with deionized water repeatedly and followed by drying at 100 °C for 1 h.
10, and calcinated in an alumina crucible at 1150 °C for 10 h in air. The obtained white powders were subsequently separated from the flux by washing with deionized water repeatedly and followed by drying at 100 °C for 1 h.
Co-catalyst loaded SrTiO3:Al particles, which we denoted as MOx/SrTiO3:Al (M = Rh, Pt, Ru, Mn, Co, Ni, Cu, RhCr, PtCr, RuCr) were prepared by impregnation of an aqueous solution of the metal precursors onto the SrTiO3:Al, followed by calcination at 350 °C for 1 h in air. SrTiO3:Al particles with 0.1 wt% of RhCrOx, which we denoted as RhCrOx (0.1 wt%)/SrTiO3:Al, was the optimum catalyst for HClO/H2 production activity, as mentioned later.
Preparation of photocatalyst film
The SrTiO3:Al particles loaded with/without co-catalyst were added to an organic vehicle consisting of a mixture of α-terpineol, 2-(2-butoxyethoxy)ethanol, and acrylic resin (SPB-TE1) with their weight ratio of 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. The weight ratio between the mixed particles and the vehicles was set to 1
2. The weight ratio between the mixed particles and the vehicles was set to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3. The obtained paste was coated onto a glass substrate (quartz) by screen-printing using a 60 μm-thick metal mask, followed by calcination in air at 300 °C for 30 min which yielded SrTiO3:Al photocatalyst films.
3. The obtained paste was coated onto a glass substrate (quartz) by screen-printing using a 60 μm-thick metal mask, followed by calcination in air at 300 °C for 30 min which yielded SrTiO3:Al photocatalyst films.
Characterization
SrTiO3:Al particles loaded with/without co-catalyst were characterized by using an X-ray diffractometer (XRD, PANalytical, X’Pert Pro, rotating anode diffractometer, 45 kV, 40 mA) with Cu Kα radiation (λKα = 1.5406 Å), a UV-vis-NIR spectrometer equipped with an integrating sphere (UV-vis DRS, Jasco, V-670), and a scanning electron microscope (SEM, HITACHI, SU-8220). X-ray photoelectron spectroscopy (XPS) was conducted using a monochromatic MgKα source (hν = 1253.6 eV) with an acceleration voltage of 8 kV and a current of 10 mA (JEOL, JPS-9000). The analysis chamber pressure was on the order of 10−6 Pa. The binding energies were calibrated using the C 1s peak (284.8 eV) as a reference.Photocatalytic reactions
In the case of the investigation of the photocatalytic HClO production on SrTiO3:Al particles loaded with/without co-catalyst, the reactions were carried out using a screw-top test tube under air. For the photocatalytic HClO and/or H2 production reaction, a Pyrex-made reaction vessel connected to a closed gas-circulating system was used. The temperature of the reaction system was controlled to keep at 25 °C using a water bath. SrTiO3:Al particles (10 mg) were added to an aqueous NaCl solution (5 mL), and light was irradiated from the side of the reactor tube by a 300 W Xe-arc lamp (Cermax PE300BF, PerkinElmer), or a simulated solar light (HAL-320, Asahi Spectra Co., Ltd.), where the light intensity was adjusted at 100 mW cm−2 (AM 1.5G).In the case of the photocatalytic HClO and/or H2 production on the SrTiO3:Al films loaded with/without co-catalyst, the reactions were carried out in a Pyrex-made reaction vessel, in which the film was horizontally fixed in 10 mL of aqueous NaCl solution, connected to a closed gas-circulating system.
The amounts of HClO produced were analyzed by using the N,N-diethyl-p-phenylenediamine (DPD) method. The detection limit of the HClO measurement was almost 0.01 μmol. The amounts of H2 produced were analyzed and quantified by using an on-line gas chromatograph (GL Science; GC-3200, TCD, Ar carrier, MS-5A column).
Results and discussion
Characterization of SrTiO3:Al photocatalyst
Optical properties, crystal structure, and surface morphology of the obtained SrTiO3:Al powders with/without a co-catalyst synthesized via the flux method were characterized. As shown in Fig. S1,† UV-vis spectra of bare and co-catalyst-loaded SrTiO3:Al (RhOx, CrOx and RhCrOx-loaded ones are shown as examples) exhibit photoabsorption below 400 nm, attributed to the inter-band transition of SrTiO3, while the broad absorption in the visible light region is due to RhCrOx species in the RhCrOx-loaded one. XRD patterns of the SrTiO3:Al samples were identified as belonging to the cubic perovskite structure, indicating that aluminum is homogeneously doped in the lattice (Fig. S2†). SEM images show that the SrTiO3:Al samples consist of angular-shaped particles with sub-micrometer diameters (Fig. S3†). These confirmed that the optical, crystalline, and morphological features were similar to those reported previously.10Photocatalytic HClO and H2 production on SrTiO3:Al photocatalyst in saline water
We first screened the co-catalyst (MOx) loading in order to identify whether or not MOx/SrTiO3:Al catalyzed the photo-oxidation reaction of Cl− in an aqueous NaCl solution. Various metal oxides such as single metal oxides (RhOx, PtOx, RuOx, MnOx, CoOx, NiOx, CuOx), and mixed metal oxides including Cr (RhCrOx, PtCrOx, RuCrOx), known for accelerating photocatalytic overall water splitting,34–38 were chosen as a co-catalyst. The photocatalytic oxidative HClO generation over a SrTiO3:Al photocatalyst loaded with/without MOx was carried out in an aqueous NaCl solution (0.5 M) under simulated solar light (AM 1.5G). The amount of produced HClO on each photocatalyst is summarized in Table 1.| No. | Co-catalyst | Amount of produced HClO (μmol) |
|---|---|---|
| a Catalyst, 0.01 g; reactant solution, 5 mL of 1 M aqueous NaCl solution; light source, simulated solar light (AM 1.5G); irradiation time, 5 h. | ||
| 1 | None | n.d. |
| 2 | Rh | 0.06 |
| 3 | Pt | 0.02 |
| 4 | Ru | 0.03 |
| 5 | Cr | n.d. |
| 6 | Mn | n.d. |
| 7 | Co | n.d. |
| 8 | Ni | n.d. |
| 9 | Cu | n.d. |
| 10 | RhCr | 4.25 |
| 11 | PtCr | 0.09 |
| 12 | RuCr | 0.10 |
The bare SrTiO3:Al exhibited almost no HClO production activity in an aqueous NaCl solution under solar light irradiation. In the case of loading a single metal oxide co-catalyst, RhOx, PtOx, and RuOx were found to slightly promote HClO production. These precious metal oxides have been reported to be active components as a co-catalyst for photocatalytic H2 evolution from water.34,38 In contrast, the tested transition metal oxides (CrOx, MnOx, CoOx, NiOx, and CuOx), mainly recognized as water oxidation co-catalysts,34,38 did not show HClO evolution activity. In all cases when cutting UV light off using an L-42 filter, HClO was almost not produced over all the samples (not shown). Also, it is generally known that HClO gradually decomposes under UV light irradiation.39 These results confirm that the photocatalytic HClO generation on SrTiO3:Al in an aqueous NaCl solution under solar light irradiation preferentially proceeds against the UV decomposition of HClO by loading appropriate co-catalysts that can especially promote H+ reduction to produce H2.
Next, photocatalytic HClO evolution activity of SrTiO3:Al powders co-loaded with the precious metal oxides and CrOx by simultaneous impregnation was tested. Notably, the existence of loaded co-catalysts (RhCrOx, PtCrOx, and RuCrOx) was confirmed by XPS (Fig. S4†), while the peaks of loaded co-catalysts were undetectable by X-ray diffraction (Fig. S5†). In addition, SEM analysis of the co-catalysts loaded SrTiO3:Al revealed the formation of nanoparticulate RhCrOx, PtCrOx, and RuCrOx (ca. 10–50 nm) over the surface of the SrTiO3:Al photocatalyst (Fig. S6†). As shown in Table 1, all the photocatalytic activity was markedly improved by co-loading with Cr. Specifically, RhCrOx-loaded SrTiO3:Al photocatalyst was found to show the best activity.
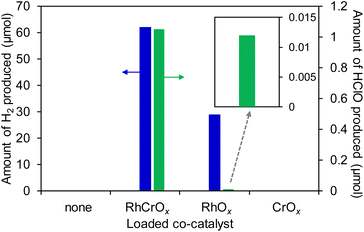
To investigate the role of the RhCrOx co-catalyst on the HClO production reaction in aqueous NaCl solution under solar-light irradiation, we conducted a series of control experiments using the RhCrOx/SrTiO3:Al photocatalyst, along with loading RhOx or CrOx alone. Prior to the investigation, the presence of the co-catalyst loaded on the SrTiO3:Al photocatalyst was confirmed by X-ray photoelectron spectroscopy (XPS). In the XPS spectra of the MOx/SrTiO3:Al, peaks attributed to the MOx (Rh 3d and Cr 2s) and the SrTiO3:Al (Sr 3p, Ti 2p, and Al 2p) appeared (Fig. S7†). However, peaks originating from the loaded MOx were undetectable by X-ray diffraction (Fig. S2†), because their amounts were too little. Fig. 1 shows the amounts of HClO and/or H2 production on the RhOx, CrOx, and RhCrOx loaded SrTiO3:Al photocatalysts in aqueous NaCl solution under solar-light irradiation. Loading of RhCrOx was the most effective to produce HClO, along with H2. The sample loaded with RhOx only, a typical reduction catalyst, also produced H2 and HClO, while the amount of HClO generated was significantly decreased compared to the case of using the RhCrOx-loaded one. In contrast, almost no product was formed in the case of using CrOx, known as an oxidation catalyst.34,38 Judging from the above-mentioned results, it is likely that the loading of a reduction co-catalyst for a counterpart H2 evolution is necessary to proceed the oxidative HClO production reaction. Indeed, in the photocatalytic reaction, O2 gas evolved preferentially compared to HClO in the oxidation reaction, and the molar ratio of O2 to H2 was near 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 exhibiting close to stoichiometric water splitting (data not shown). For example, the selectivity for HClO production on the RhCrOx/SrTiO3:Al photocatalyst was 1.8%. It indicates that HClO is an oxidative by-product in the present conditions because the SrTiO3:Al photocatalysts were originally designed for overall water splitting. Improving the selectivity will be examined in future work by further optimization of the co-catalyst modification.
2 exhibiting close to stoichiometric water splitting (data not shown). For example, the selectivity for HClO production on the RhCrOx/SrTiO3:Al photocatalyst was 1.8%. It indicates that HClO is an oxidative by-product in the present conditions because the SrTiO3:Al photocatalysts were originally designed for overall water splitting. Improving the selectivity will be examined in future work by further optimization of the co-catalyst modification.
Next, in order to investigate the reason for the lower HClO production activity on RhOx/SrTiO3:Al, the following HClO decomposition reaction tests were conducted.40 It is generally known the decomposition reaction of HClO proceeds on the metal oxide surface,40 following the equation (eqn (4)):
| 2ClO− → 2Cl− + O2 | (4) |
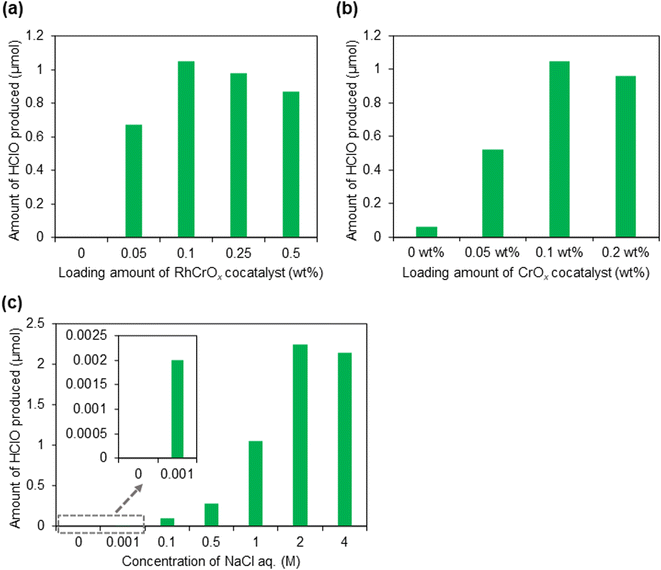
Initial screening of the co-catalysts revealed that the loading of RhCrOx was found to boost the photocatalytic HClO production in aqueous NaCl solution. We subsequently conducted a more detailed investigation of the catalyst composition. The HClO/H2 production on the SrTiO3:Al photocatalysts loaded with different amounts of RhCrOx was examined under simulated solar light irradiation. The photocatalytic activity of the RhCrOx/SrTiO3:Al photocatalyst in the HClO/H2 production reaction depended on the loading amount of RhCrOx as shown in Fig. 2(a), increasing the loading amount up to 0.1 wt% resulted in an increase in the activity, whereas beyond 0.25 wt% the activity decreased. The promotion of HClO/H2 generation by loading RhCrOx can be caused by an efficient charge separation between SrTiO3:Al and the co-catalyst and the inactivity with HClO of the surface CrOx species as mentioned above. However, the excessively loaded RhCrOx co-catalyst may cover the surface active sites of the photocatalysts and hinder its contact with the reaction substrates (water and Cl−), and/or shield the incident light that should be absorbed by the photocatalyst, resulting in the decreased activity. Alternatively, the amounts of HClO produced on the SrTiO3:Al photocatalysts loaded with RhCrOx were also dependent on the Cr loading amount, 0.1 wt% of Cr loaded sample exhibited the highest activity in the photocatalytic HClO production (Fig. 2(b)).
We also examined the effect of the NaCl concentration in the solution on the HClO/H2 production reaction over the RhCrOx/SrTiO3:Al photocatalyst under the simulated solar-light irradiation (Fig. 2(c)). The activity of HClO generation increased with increasing NaCl concentration up to 2 M, whereas a concentration of NaCl more than 2 M did not affect the activity. The result indicates that the concentration of the reactant Cl− is important to proceed the reaction smoothly. It is noted that the HClO was produced even when the NaCl concentration was <0.5 M, suggesting the possibility of the practical application using seawater (Cl− concentration: ca. 0.5 M), or tap water (Cl− concentration: ca. <0.01 M).
Additionally, we conducted a recycle test using the present RhCrOx/SrTiO3:Al particles. The RhCrOx/SrTiO3:Al particles were separated from the reaction solution, washed with aqueous solution, and dried at 80 °C. The photooxidative HClO production reaction in aqueous NaCl solution was conducted using the obtained RhCrOx/SrTiO3:Al particles. These processes were repeated 2 times. From the characterizations of the RhCrOx/SrTiO3:Al photocatalyst before/after the reaction shown in Fig. S9,† no obvious changes were observed in the original particles and co-catalyst characteristics. In addition, the photocatalytic HClO production ability of the RhCrOx/SrTiO3:Al particles was almost not changed over at least 3 cycles (Fig. S10†), indicating the stability of the RhCrOx/SrTiO3:Al photocatalyst towards these photooxidative conditions.
Photocatalytic HClO and H2 production on SrTiO3:Al films in saline water
Alternatively, photocatalyst films were prepared via screen-printing and subsequent drying and calcination (at 300 °C) using the best photocatalyst, RhCrOx (0.1 wt%)/SrTiO3:Al, toward constructing a module system of efficient solar HClO/H2 production. A photograph of the RhCrOx/SrTiO3:Al film is shown in Fig. 3(a). It was confirmed that there were no obvious differences between the RhCrOx/SrTiO3:Al particles and the film in the XRD patterns and the UV-vis diffuse reflection spectra (Fig. S11†). Fig. 3(b and c) shows the SEM images of the RhCrOx/SrTiO3:Al film. As seen in the cross-section views (b), the RhCrOx/SrTiO3:Al film possessed a homogeneous thickness of ca. 10 μm with flat surfaces. The magnified cross-section view (c) revealed that the film has a densely packed porous structure. From the top view, the RhCrOx/SrTiO3:Al film was confirmed to consist of particles with sizes of ca. 300–500 nm. | ||
| Fig. 3 (a) A photograph of RhCrOx/SrTiO3:Al film prepared via the screen-printing method. (b) Cross-section and (c) magnified cross-section views of SEM images of the RhCrOx/SrTiO3:Al film. | ||
Firstly, we checked the HClO and H2 production ability in an aqueous NaCl solution on the obtained RhCrOx/SrTiO3:Al film under UV light irradiation. Under UV-light irradiation, gas bubbles were obviously observed, as shown in Fig. S12(a),† and the color of the reaction solution changed to pink when the DPD reagent was added, indicating generation of HClO (b). The produced amount of HClO was 7.5 μmol cm−2 under UV-light irradiation for 1 hour. H2 and HClO production activity on the RhCrOx/SrTiO3:Al film was also evaluated under simulated solar light. Fig. 4 shows the time course of gas evolution on the RhCrOx/SrTiO3:Al film. At the early stages of photoirradiation, both H2 and HClO were generated simultaneously with steady rates from the aqueous NaCl solution. However, the rate of HClO generation slightly decreased as the irradiation time increased. This is because the generated HClO is subsequently decomposed on the surface of the co-catalyst, as mentioned above. The H2 and HClO production activity on the RhCrOx/SrTiO3:Al film was almost the same as that on the corresponding suspended particles (Fig. S13†). The result indicates that the porous structure in the present RhCrOx/SrTiO3:Al film enables an efficient mass transfer of the reaction substances, and thereby contributes to maintaining the original activity of the photocatalyst particles for H2 and HClO production.
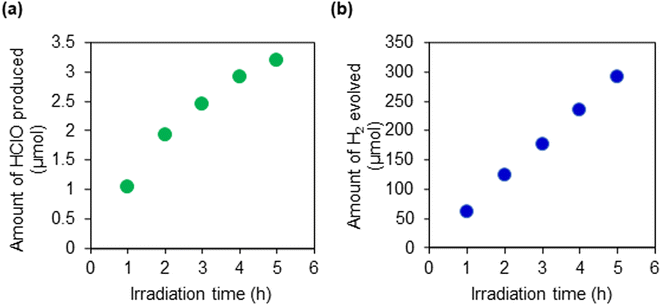 | ||
| Fig. 4 The time course of (a) HClO and (b) H2 evolution on the RhCrOx/SrTiO3:Al film (active area: 6.25 cm2) in an aqueous NaCl solution (1 M) under simulated solar light (AM 1.5G). | ||
Conclusions
Solar-light driven simultaneous H2/HClO production from saline water was demonstrated for the first time using RhCrOx/SrTiO3:Al photocatalyst systems. In this reaction, loading co-catalyst onto the surface of the SrTiO3:Al photocatalyst significantly affected the HClO production activity; the loading of Rh, Pt, Ru which are known to promote the proton reduction reaction, improved the HClO production. Mixing Cr into those co-catalysts further improved the activity, particularly the RhCrOx-loaded SrTiO3:Al photocatalyst exhibited the highest activity. It is found that CrOx effectively suppresses the decomposition of produced HClO whereas Rh alone does not in the RhCrOx. The activity of photocatalytic HClO production over RhCrOx/SrTiO3:Al was influenced by the reactant Cl− concentration, improving as it increased.Toward the practical use of solar H2/HClO production, RhCrOx/SrTiO3:Al photocatalyst panel systems, wherein the photocatalyst particles are fixed onto a glass substrate, were also developed. A porous RhCrOx/SrTiO3:Al photocatalyst film with a thickness of 10 μm was prepared via simple screen printing followed by a drying and calcination process. The obtained film produced H2 and HClO from saline water under simulated solar light irradiation, almost maintaining the same activity as on corresponding suspended particles.
For practical applications of solar H2 and/or HClO production using saline water, it is essential to develop photocatalyst particles/films capable of producing them efficiently under solar-light irradiation. Such developments will be vital to upgrade photocatalytic materials to improve various factors such as photoabsorption or selectivity of HClO production. We believe that this work will contribute to the development of sustainable and economical solar energy conversion systems that can obtain both clean energy and high-value-added chemicals.
Author contributions
Sayuri Okunaka: investigation, resources, writing – original draft, writing – review & editing. Toshio Nakamura: investigation, writing – review. Takeshi Ikeda: SEM observation, writing – review. Kohei Tsuruda: XPS measurement, writing – review. Hiromasa Tokudome: conceptualization, writing – original draft, writing – review & editing, supervision.Conflicts of interest
There are no conflicts to declare.Notes and references
- S. Chen, T. Takata and K. Domen, Nat. Rev. Mater., 2017, 2, 17050 CrossRef CAS.
- D. Voiry, H. S. Shin, K. P. Loh and M. Chhowalla, Nat. Rev. Chem, 2018, 2, 0105 CrossRef CAS.
- Y. Wang, X. Shang, J. Shen, Z. Zhang, D. Wang, J. Lin, J. C. S. Wu, X. Fu, X. Wang and C. Li, Nat. Commun., 2020, 11, 3043 CrossRef CAS PubMed.
- X.-B. Li, C.-H. Tung and L.-Z. Wu, Nat. Rev. Chem, 2018, 2, 160–173 CrossRef CAS.
- D. M. Fabian, S. Hu, N. Singh, F. A. Houle, T. Hisatomi, K. Domen, F. E. Osterloh and S. Ardo, Energy Environ. Sci., 2015, 8, 2825–2850 RSC.
- Q. Wang and K. Domen, Chem. Rev., 2020, 120, 919–985 CrossRef CAS PubMed.
- F. E. Osterloh, ACS Energy Lett., 2017, 2(2), 445–453 CrossRef CAS.
- K. Takanabe, ACS Catal., 2017, 7, 8006–8022 CrossRef CAS.
- Y. Wang, H. Suzuki, J. Xie, O. Tomita, D. J. Martin, M. Higashi, D. Kong, R. Abe and J. Tang, Chem. Rev., 2018, 118, 5201–5241 CrossRef CAS PubMed.
- Y. Goto, T. Hisatomi, Q. Wang, T. Higashi, K. Ishikariyama, T. Maeda, Y. Sakata, S. Okunaka, H. Tokudome, M. Kayatama, S. Akiyama, H. Nishiyama, Y. Inoue, T. Takewaki, T. Setoyama, T. Minegishi, T. Takata, T. Yamada and K. Domen, Joule, 2018, 2, 509–520 CrossRef CAS.
- J. H. Kim, D. Hansora, P. Sharma, J. Jang and J. Lee, Chem. Soc. Rev., 2019, 48, 1908–1971 RSC.
- T. Takata, K. Domen, S. Okunaka, H. Tokudome, N. Kato, T. Morikawa and Y. Takeda, in Recent Developments in Functional Materials for Artificial Photosynthesis, ed. S. Ghosh and Q. Wang, The Royal Society of Chemistry, 2023, ch. 12, pp. 331–362 Search PubMed.
- A. Xiong, G. Ma, K. Maeda, T. Takata, T. Hisatomi, T. Setoyama, J. Kubota and K. Domen, Catal. Sci. Technol., 2014, 4, 325 RSC.
- S. Okunaka, H. Tokudome and R. Abe, Catal. Sci. Technol., 2016, 6, 254 RSC.
- S. Okunaka, H. Tokudome and R. Abe, Chem. Lett., 2016, 45, 57 CrossRef CAS.
- Q. Wang, T. Hisatomi, Q. Jia, H. Tokudome, M. Zhong, C. Wang, Z. Pan, T. Takata, M. Nakabayashi, N. Shibata, Y. Li, I. D. Sharp, A. Kudo, T. Yamada and K. Domen, Nat. Mater., 2016, 15, 611 CrossRef CAS PubMed.
- S. Okunaka, H. Kameshige, T. Ikeda, H. Tokudome, T. Hisatomi, T. Yamada and K. Domen, ChemSusChem, 2020, 13, 4906 CrossRef CAS PubMed.
- S. Nandy, T. Hisatomi, M. Nakabayashi, H. Li, X. Wang, N. Shibata, T. Takata and K. Domen, Joule, 2023, 7, 1641–1651 CrossRef CAS.
- H. Nishiyama, T. Yamada, M. Nakabayashi, Y. Maehara, M. Yamaguchi, Y. Kuromiya, Y. Nagatsuma, H. Tokudome, S. Akiyama, T. Watanabe, R. Narushima, S. Okunaka, N. Shibata, T. Takata, T. Hisatomi and K. Domen, Nature, 2021, 598, 304 CrossRef CAS PubMed.
- Y. Shiraishi, Y. Ueda, A. Soramoto, H. Hinokuma and T. Hirai, Nat. Commun., 2020, 11, 3386 CrossRef CAS PubMed.
- K. Sayama, ACS Energy Lett., 2018, 3(5), 1093–1101 CrossRef CAS.
- K. Fuku and K. Sayama, Chem. Commun., 2016, 52, 5406–5409 RSC.
- K. Fuku, N. Wang, Y. Miseki, T. Funaki and K. Sayama, ChemSusChem, 2015, 8, 1593–1600 CrossRef CAS PubMed.
- S. Iguchi, Y. Miseki and K. Sayama, Sustainable Energy Fuels, 2018, 2, 155–162 RSC.
- R. Pan, Y. Miseki, S. Okunaka and K. Sayama, ACS Sustainable Chem. Eng., 2020, 8(23), 8629–8637 CrossRef.
- S. Okunaka, Y. Miseki and K. Sayama, Catal. Sci. Technol., 2021, 11, 5467–5471 RSC.
- S. Okunaka, Y. Miseki and K. Sayama, J. Ceram. Soc. Jpn., 2022, 130, 395–402 CrossRef CAS.
- R. Pan, Y. Miseki and K. Sayama, Catal. Sci. Technol., 2022, 12, 2935–2942 RSC.
- Y. Shiraishi, Y. Shimabukuro, K. Shima, S. Ichikawa, S. Tanaka and T. Hirai, JACS Au, 2023, 3(5), 1403–1412 CrossRef CAS PubMed.
- W. A. Rutala and D. J. Weber, Clin. Microbiol. Rev., 1997, 10, 597–610 CrossRef CAS PubMed.
- L. Wang, M. Bassiri, R. Najafi, K. Najafi, J. Yang, B. Khosrovi, W. Hwong, E. Barati, B. Belisle, C. Celeri and M. C. Robson, J. Burns Wounds, 2007, 6, 65–79 Search PubMed.
- T. Takata, J. Jiang, Y. Sakata, M. Nakabayashi, N. Shibata, V. Nandal, K. Seki, T. Hisatomi and K. Domen, Nature, 2020, 581, 411 CrossRef CAS PubMed.
- C. Avcıoğlu, S. Avcıoğlu, M. F. Bekheet and A. Gurlo, ACS Appl. Energy Mater., 2023, 6(3), 1134–1154 CrossRef.
- K. Maeda and K. Domen, Bull. Chem. Soc. Jpn., 2016, 89, 627–648 CrossRef CAS.
- J. Yang, D. Wang, H. Han and C. Li, Acc. Chem. Res., 2013, 46(8), 1900–1909 CrossRef CAS PubMed.
- K. Maeda, K. Teramura, D. Lu, N. Saito, Y. Inoue and K. Domen, Angew. Chem., Int. Ed., 2006, 45, 7806–7809 CrossRef CAS PubMed.
- K. Maeda, K. Teramura, D. Lu, N. Saito, Y. Inoue and K. Domen, J. Phys. Chem. C, 2007, 111, 7554–7560 CrossRef CAS.
- T. Hisatomi, K. Takanabe and K. Domen, Catal. Lett., 2015, 145, 95–108 CrossRef CAS.
- M. J. Watts and K. G. Linden, Water Res., 2007, 41, 2871–2878 CrossRef CAS PubMed.
- M. Abe, J. Magn. Soc. Jpn., 1998, 22, 1225 CAS.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d3se01163a |
| ‡ Present address: Department of Applied Chemistry, Faculty of Science and Engineering, Tokyo City University, 1-28-1 Tamazutsumi, Setagaya-ku, Tokyo, 158-8557, Japan. E-mail: E-mail: okunakas@tcu.co.jp |
| This journal is © The Royal Society of Chemistry 2024 |