A solid-state Li–air battery: computational studies of interfaces and relevance to discharge mechanism
Abstract
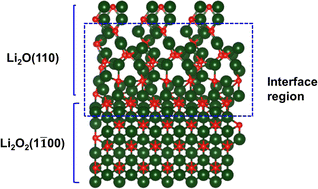
There is much interest in developing new energy storage systems to replace currently available ones that mainly work based on Li-ion intercalations. One attractive area is the Li–air battery for which most of the research has involved liquid electrolytes. There have been few studies on the use of a solid electrolyte in a Li–air battery. Recently, we reported the successful use of a solid-state electrolyte in a Li–air battery resulting in a Li2O product and potentially much higher energy density than in a Li–air battery based on either a Li2O2 or LiO2 product (Science, 2023, 379, 499). In this paper we discuss how the discharge mechanism involved in this solid-state Li–air battery differs from that of a Li–air battery with a liquid electrolyte. The solid-state mechanism is further explored with density functional studies of various interfaces involving the discharge product. We discuss the relevance of the results to the discharge mechanism in the solid-state Li–air battery.
- This article is part of the themed collection: Rechargeable non-aqueous metal-oxygen batteries


 Please wait while we load your content...
Please wait while we load your content...