A dynamic anode boosting sulfamerazine mineralization via electrochemical oxidation†
Abstract
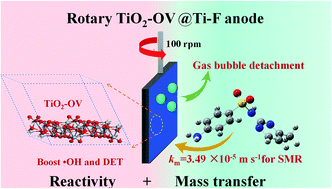
Despite numerous efforts to treat wastewater with sulfonamides, their mineralization has rarely been achieved, resulting in the generation of more toxic by-products. In this study, greater than 75% mineralization of sulfamerazine (SMR) was achieved following 4 h of electrochemical oxidation. Remarkably, the Microtox® toxicity test confirmed the elimination of by-products with higher toxicity. The electrochemical treatment process was carried out using a dynamic oxygen-vacancy-mediated TiO2 anode (TiO2-OV@Ti-F), which comprised oxygen-deficient Ti4O7 coated on titanium-foam (Ti-F) via thermal spraying, allowing simultaneous high reactivity and mass transfer. SMR degradation followed a pseudo-first-order kinetics model, where the rate constant (kapp = 1.64 × 10−2 min−1) for the rotary TiO2-OV@Ti-F configuration was 1.98-fold greater than that of the static one (kapp = 8.30 × 10−3 min−1). This highlights the superiority of the rotary TiO2-OV@Ti-F anode for SMR decay. The high oxidation capabilities arose from: (i) the synergetic effect between the rotating system and the Ti4O7 coating; (ii) the enhanced mass transfer coefficient (3.49 × 10−5 m s−1) in the rotating configuration, as well as the increase in SMR degradation via direct oxidation, due to a low hole injection energy, as supported by density functional theory calculations; and (iii) boosted ˙OH formation achieved via removing the gas bubbles attached to the anode, along with lower adsorption energies for H2O and ˙OH. The results revealed that rotary TiO2-OV@Ti-F is a promising alternative for antibiotic wastewater treatment owing to its high organic mineralization and low level of energy consumption (0.29 kW per h per gTOC).


 Please wait while we load your content...
Please wait while we load your content...